Pest & Crop Newsletter, Entomology Extension, Purdue University
- September Surprise, Soybean Aphid Continues in Southern Counties
- Harvest Restrictions for Soybean Aphid Insecticides
- Updated Map: Known Counties With Western Bean Cutworm Infestations
- Trochanter Mealybug in Southern Indiana
- Complacency Increases Helps to Spread Hessian Fly
- Black Light Trap Catch Report
- Cool Temperatures Impact on Soybean Yield and Maturity (Sept. 11)
- Wheat Planting Considerations: Impacts of 2009 Crop Harvests (Sept. 16)
- Yield Monitor Calibration: Garbage In, Garbage Out
- Reddish-Purple Corn Plants Late in the Season
- Corn Ear Damage Caused by Bird Feeding
September Surprise, Soybean Aphid Continues in Southern Counties- (Christian Krupke)
No sooner did we sound an “all clear” on aphids for most of the state, than did we hear of high populations in the south and particularly southwestern parts of Indiana. Colleagues in adjacent areas of Kentucky and Ohio are reporting similar infestations. These are some of the latest-planted beans and so are the “only game in town” for hungry aphids seeking nutrition.
The problem with this unprecedented late infestation is that growers are asking: “Do I treat, even through the beans are close to R6?” Unfortunately, we don’t have much to go by here – we haven’t had a late surge in aphids of this magnitude before, so we have no good research data to point to. Bear in mind that the 250-aphid threshold was developed primarily to target early R-stage infestations and be conservative (minimize risk). That threshold is even more conservative for an older plant. So while an R5 bean can undoubtedly withstand larger numbers than an R2 plant, we don’t know exactly how high aphids need to be to be worth spraying – but when the costs of treatment, (including destroying plants with spray tracks) are figured into the equation, treating aphid infestations at or near the threshold is probably not economic in mid to late R5 beans.
Keep an eye out in your late beans – especially down south (although we have reports of high aphids from the northwestern county on extremely late beans as well). Many winged aphids on leaves are also an indication that these individuals will soon leave the field and migrate, likely to buckthorn – their overwintering host.
![]()
Harvest Restrictions for Soybean Aphid Insecticides - (Christian Krupke and John Obermeyer)
The following listing includes the insecticides registered for soybean aphid, including rate per acre and harvest restrictions. Refer to the label specific rates and application information.
| Soybean Aphid Insecticides and Harvest Restrictions | ||
| Product | Rate and Formulation | Days Before Harvest |
| chlorpyrifos (Lorsban)* | 2 pt. 4E | 28 |
| chlorpyrifos & gamma cyhalothrin (Cobalt)* | 13-26 fl. oz. EC | 30 |
| cyfluthrin (Baythroid XL)* | 2-2.8 oz. 3EC | 45 |
| deltamethrin (Delta Gold)* | 1.5-19 fl. oz. 1.5EC | 21 |
| esfenvalerate (Asana XL)* | 5.8-9.67 oz. 0.66EC | 21 |
| gamma-cyhalothrin (Proaxis)* | 1.9-3.2 oz. 0.5EC | 45 |
| imidacloprid & cyfluthrin (Leverage)* | 3.8 fl oz. SE | 45 |
| lambda-cyhalothrin (Warrior)* | 1.9-3.2 oz. 1CS | 30 |
| zeta-cypermethrin (Mustang Max)* | 2.8-4.0 oz. 0.8EW | 21 |
| zeta-cypermethrin & bifenthrin (Hero)* | 4.0-10.3 oz. EC | 21 |
| *Restricted Use Pesticide | ||
![]()
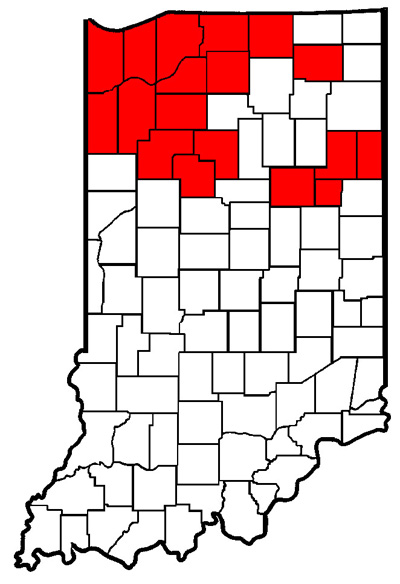
Updated Map: Known Counties With Western Bean Cutworm Infestations - (John Obermeyer)
Thanks to those keeping us updated on where western bean cutworm larvae and damage have been found. As you can see below, most of the northern counties have reported this pest. Should you have an addition to the following county map, please email (attach pictures if you have them) to: <obe@purdue.edu>. Thanks!
Western bean cutworm found in the counties marked red.
Western bean cutworm pre-pupa within its earthen cell.
Western bean cutworm pre-pupa, next to knife blade, found 9 inches deep in this sandy soil.
![]()
Trochanter Mealybug in Southern Indiana - (Christian Krupke and Larry Bledsoe)
A new insect to watch for, and potential pest, has surfaced in southern Indiana soybeans. The trochanter mealybug, Pseudococcus sorghiellus, was first found in Kentucky last year and has also been found in Ohio. Similar to other mealybugs in appearance, these small, whitish insects live beneath the soil surface and feed on plant juices. The above-ground symptoms are similar to K-deficiency – yellowing of foliage and stunting. If you have beans that exhibit these symptoms, we would recommend digging up a sample and shaking the roots over dark-colored paper. The small, grayish-white insects will soon begin crawling around the paper. A hand lens may be required to confirm diagnosis.
We don’t know much more about them at this point – are they the primary cause of the yellowing, or are they attacking previously-stressed plants? We are hoping to get an idea of how common these insects are throughout the state, and whether geography, soil type, maturity etc., play significant roles. We are in the earliest stages of finding out about this insect, so any information you could provide would be helpful.
This find was reported by consultant Greg Weaver, with photos provided by Shelby county ANR educator Scott Gabbard.
Trochanter mealybug attached to soybean root. (Photo Credit: Scott Gabbard, Shelby County ANR Educator)
![]()
Complacency Increases Helps to Spread Hessian Fly – (Brandi Schemerhorn and Sue Cambron)
- Remember to utilize fly free dates.
- Destruction of volunteer wheat helps reduce insect reservoir to avoid spring infestations.
The Hessian fly is present in wheat growing areas throughout the US, including Indiana. Even if wheat is not planted in a particular area, the Hessian fly can survive on alternative grass hosts. When the opportunity presents itself for a wheat infestation, there is potential for rapid increase of fly populations as a result of weather conditions or cropping practices that favor survival of eggs and young larvae in the fall.
Following the fly-free date.
A low fall infestation often goes unnoticed due to the tillering of the wheat plant. Much of the fall fly population can be avoided by planting after the fly-free date. The fly-free date is of key importance, even if you plan to use the wheat only for cattle. This fly-free date is our main protection to avoid a subsequent infestation by the spring brood. Additionally, it has been shown that following the fly-free date will help reduce other wheat disease problems and reduce winter-kill from excessive growth. Crop rotation is a key management strategy for reducing Hessian fly problems.
Test plots showing resistant and susceptible wheat.
Plowing fields after wheat harvest destroys the fly.
The Hessian fly passes the summer in the stubble of the current wheat crop. Volunteer wheat germinates and begins growing just in time for the fall emergence of the Hessian fly. These plants are readily infested resulting in a rapid build-up of the population. Removal of volunteer wheat before the emergence of the fall brood greatly reduces the insect reservoir for a spring infestation.
Infestations were heavy in southern Illinois and Missouri this year, but reports of Hessian fly damage in Indiana was down due greatly to the adherence to the fly free dates. Wisconsin is now considering a law to require adherence by farmers to the fly free dates.
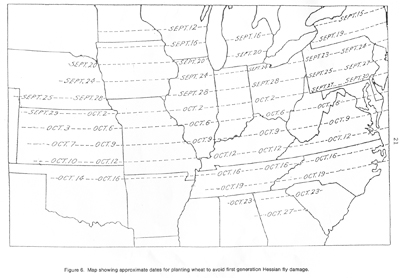
Map showing approximate dates for planting wheat to avoid first generation Hessian fly damage
![]()
Click here to view the Black Light Trap Catch Report
Dealing with Diplodia Ear Rot - (Kiersten Wise and Charles Woloshuk)
Scouting reports across Indiana indicate that Diplodia ear rot is present in Indiana fields. Many areas of Indiana had wet weather at and during silking, which favors infection by the fungus causing Diplodia ear rot, Stenocarpella maydis. In susceptible hybrids, substantial yield losses can occur. The fungus can infect plants through the husk or the shank, and rot the ear before corn is fully mature. Infected ears often have bleached husks with tiny black specks, which are the spore producing pycnidia. Removal of the husk of an infected ear will reveal white fuzzy growth of the pathogen between the kernels, which often starts at the base of the ear (Figure 1). The cob can also appear rotted. In the US, mycotoxins have not been associated with Diplodia ear rot; however, feeding value and storability of the grain is reduced with infection. It is important to properly store grain damaged by Diplodia ear rot. Drying grain to 15 percent moisture after harvest and cooling and storing infected grain below 50°F will prevent further growth of the fungus. The best way to manage Diplodia ear rot is to avoid planting susceptible hybrids, and avoid corn-on-corn rotations or planting into corn residue. For more information on Diplodia ear rot, please read the Purdue Extension bulletin, Diplodia ear rot at: <http://www.extension.purdue.edu/extmedia/BP/BP-75-W.pdf>
Figure 1. Diplodia ear rot on corn (Photo Courtesy: G. Shaner).)
Cool Temperatures Impact on Soybean Yield and Maturity (Sept. 11) - (Shaun Casteel)
Fall officially arrives in two weeks, but we have felt the fall weather for what seems like two months across most of Indiana. The combination of delayed planting and decreased heat unit accumulation raises concerns for the yield and the maturity of the soybean crop.
Yield of First-Crop Soybean
The late planting of soybean reduced the probability of attaining the maximum yield in a normal year (see Figure 3, <http://www.extension.entm.purdue.edu/pestcrop/2009/issue11/index.html#delayed>). Favorable environmental conditions—timely precipitation, good sunlight, and heat unit accumulation—could have provided the ingredients to recover from late planting. However, cool temperatures in July added insult to injury and probably reduced the number of flowers initiated and pods retained, especially in the late-planted soybeans. Flower initiation, pod set, and pod retention was reduced in a study of a determinate soybean that was exposed to 50°F for one week at various reproductive stages (Musser et. al., 1983b). Fortunately, the indeterminate soybean grown in the Midwest provides some flexibility and adaptability for flowering and pod set. Many areas in the northern third of Indiana were planted in a timely fashion, but received little precipitation in July. The severity of the water stress was minimal in these areas due to the sluggish plant growth (vegetative and reproductive) that resulted from cool temperatures. The yields for this year will not be bin busters, but the weather forecast for the following two weeks is promising for seed fill in first-crop soybeans.
Maturity of First-Crop Soybean
Late plantings have historically delayed maturity (and ultimately harvest) on a ratio of 3 to 1. Many fields across Indiana were planted three weeks late, which would delay maturity by approximately one week. Cool temperatures during reproductive growth have probably caused greater delays in maturity. For example, chilling (50°F for one week) during reproductive initiation of a determinate soybean delayed anthesis by one week (Musser et. al., 1983b). The combination of late planting and cool temperatures during reproductive stages has probably delayed the soybean crop 7 to 10 days in the northern third and 14 to 21 days in the central and southern areas compared to a “normal” year.
The majority of first-crop soybeans are at R5 (initiation of seed) to R6 (full seed) (Figure 1). An average soybean at R5 and R6 will mature to R7 (initiation of physiological maturity) in approximately 33 and 18 days, respectively, and could be prolonged with cool temperatures. However, the weather forecast in the coming weeks calls for daily highs in the upper 70s to low 80s and daily lows in the 50s. Soybean maturation should progress steadily over the coming weeks. In fact, late maturity group 2 soybeans are senescing and dropping leaves in west-central and north-central Indiana. Soybean harvest will be in full swing in October, which raises some concerns for planting wheat and these implications will be addressed in a follow-up article.
Figure 1. R5 is the initiation of seed, and the soybean seed is 1/8 of an inch long within a pod at one of the four uppermost nodes on the main stem. R6 is the full seed growth stage, and the soybean seed fills the pod cavity at one of the four uppermost nodes on the main stem.
Yield and Maturity of Double-Crop Soybean
Many producers in the northern and central regions of Indiana rolled the dice and planted double-crop soybeans near the planting cutoff date (anecdotally determined to be 90 days prior to the first 32°F frost). The southern regions of Indiana have a wider window for adequate heat unit accumulation prior to fall frosts and freezes than the northern and the central regions. Generally, the first fall frost of 32°F occurs around October 6 to 13 in the northern third, October 10 to 15 in the middle third, and October 14 to 23 in the southern third (~October 25 in southwestern corner) at a 50% probability (Purdue Corn and Soybean Guide, 2009). A hard freeze of 28°F occurs approximately 10 days later than the 32°F frost.
Double-crop soybean growth stages are across the board from R1 (flower initiation) to R5 (initiation of seed). The time needed for the soybeans to mature is limited, especially in the northern and central areas. For instance, double crop soybeans at R3 (pod initiation) will take approximately 50 days to reach R7, which would be October 30. This would be about 5 to 7 days after the first hard freeze of 28°F in the northern and central areas of Indiana. Seeds in green pods (~65% moisture) were injured at 28°F; whereas, seeds in brown pods (~35% moisture) were not injured at 10°F (Judd et. al., 1982). The risk for seed injury, seed quality degradation, and yield loss decreases as soybeans move from R6 to R7.
Literature Cited:
Judd, R., D.M. Tekrony, D.B. Egli, and G.M. White. 1982. Effect of freezing temperatures during soybean seed maturation on seed quality. Agron. J. 74:645-650.
Musser, R.L., S.A. Thomas, J.F. Thomas, and P.J. Kramer. 1983. Effects of shoot chilling during floral initiation and development of “Ransom” soybeans. Plant Physiol. Suppl. 72:43.
Purdue University Corn and Soybean Guide 2009 edition (ID-179) p. 279-282.
![]()
Wheat Planting Considerations: Impacts of 2009 Crop Harvests (Sept. 16) - (Shaun Casteel)
Planting wheat in Indiana usually depends on three main factors: Hessian fly-free date, harvest of corn and soybean, and the weather. Weather conditions were just right for head scab infection in many wheat fields this spring and could reduce seed quality. The wet spring and cool temperatures in July and August have delayed and potentially reduced yield of the corn and soybean crops (related articles at <http://www.kingcorn.org/cafe>. Additionally, delayed harvests of corn and soybean will certainly be of interest to planting dates of soft red wheat in Indiana.
Planting Date
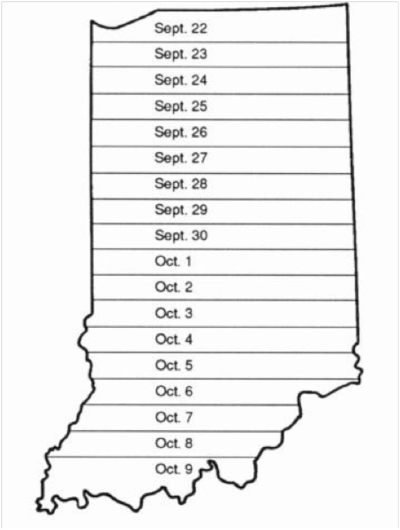
The optimal planting date for wheat in Indiana is within 7 to 14 days after the average Hessian fly-free date, which is presented in Figure 1. Early planting of wheat increases the probability of infestation by Hessian flies, which lay eggs that hatch and the larvae (maggots) feed on young wheat seedlings (for more information please visit <http://www .extension.entm.purdue.edu/fieldcropsipm/insects/hessianfly.html> The risk for disease infection in the fall increases with early planting, especially for seedling blights in warm soils (> 60°F) and barley yellow dwarf virus that is transmitted by aphids. In contrast, late-planted wheat often has limited vegetative growth (i.e., tillering) that can decrease winter survivability, increase the susceptibility of winter heaving, and reduce yield. Wheat yields may be reduced 10 to 20% if planting is delayed up to 30 days after the Hessian fly-free date.
For example, the Hessian fly-free date in Whitley County (northeastern Indiana) would be ~September 24, and wheat should ideally be planted by October 7 to minimize stresses and maximize yield potential. Some soybean fields in Whitely County are at full seed (R6) today, September 16, and will begin physiological maturity (R7) by October 4 based on average soybean development (~18 days from R6 to R7). Hence, the window for optimal planting of wheat can be short due to the delayed soybean maturity. This is a concern in the northern and central areas of Indiana. However, some combines have been cutting soybeans in Tippecanoe County and timely planting of wheat is still possible.
Figure 1. Average Hessian fly-free dates for Indiana. (Illustration by C. Mansfield and S. Hawkins, Extension Agronomist, Purdue University
Seeding Rate
Optimum yields are normally obtained at plant populations of 1.3 to 1.5 million plants per acre (30 to 35 plants per ft2). The amount of seed needed to obtain this stand would vary depending on the seed size, germination test, and emergence potential (factors include planting date, planting equipment, and seed bed conditions).
Wheat seed that has 90% germination value and the field conditions are such that 90% of the viable seed will emerge equals 81% of the planted seed will establish plants (90% germinate x 90% emerge = 81% establish). The seeding rate to obtain 1.5 million plants per acre would be 1.85 million seeds per acre, and it is calculated as follows: [(1.5 million plants per acre) / (81% plant establishment) = 1.85 million seeds per acre]. The number of pounds per acre will increase as the seed size increases, so the seeding rate of 1.85 million seeds per acre will require 115, 132, and 154 lb per acre for small (16,000 seeds per lb), medium (14,000 seeds per lb), and large seeds (12,000 seeds per lb), respectively. Plant population of 1.3 million per acre would require 1.6 million seeds per acre if 81% plant establishment was used, and small, medium, and large seeds would be planted at 100, 114, and 133 lb per acre, respectively. Seeding rate would decrease if plant establishment values increased (e.g., higher germination values, optimal soil moisture, optimal soil temperature, use of seed treatments). If you are planting in an area that is prone to winter heaving and winter kill can be problematic, you should aim for 35 plants per ft2. Late plantings of wheat should also increase the desired seeding rate to compensate for the limited time for adequate tillering.
Seed Depth
Seed placement is important for emergence, plant stand, and winter hardiness. Seeding depths around 1” are ideal, and an acceptable range is 0.75 to 1.25.” If the seeds are planted too deep, the seed uses much of its energy to emerge with little reserves left for establishment. Shallow planting enables quick emergence provided soil moisture is adequate. However, if the root system does not fully develop the crop will be susceptible to stresses in water and nutrient availability. Shallow root systems will not anchor the plants and will be prone to heaving during freezing-thawing cycles. Thus, it is critical to calibrate planting equipment (e.g., replace worn seed openers, calibrate depth control, adjust coulters to cut through crop residue) and control planting speeds to maintain seed placement.
Seed Source
Seed treatments with fungicides should prove beneficial this year as wheat plantings could be delayed. Head scab was prevalent in many wheat producing areas during the 2009 harvest, so be on the lookout for scabby wheat especially in bin-run wheat. I recommend planting certified seed to sidestep the issues of planting scabby wheat—reduced vigor, poor germination, and source of disease. Scab-infected seeds and “tombstones” will NOT germinate, and seed treatments will NOT bring them back to life. Bin-run wheat should be thoroughly cleaned to ensure purity and to reduce scabby wheat. This seed should also be tested for germination and treated with fungicides labeled for Fusarium graminearum, the causal agent of head scab, to help protect uninfected seeds from early season infection. The seed treatment will not prevent head scab next spring since head scab depends on weather conditions at the time of flowering, previous crop, and varietal susceptibility.
![]()
Yield Monitor Calibration: Garbage In, Garbage Out - (Bob Nielsen)
Grain yield monitors have been in vogue for more than 10 years and can provide valuable spatial yield information to growers. Yield monitors offer a visual diversion from the boredom of harvest. They provide a source of historical yield records more detailed than that offered by elevator weigh tickets. They provide a viable alternative to weigh wagons or farm scales for measuring yields in on-farm research trials. When connected to a DGPS receiver, yield monitors generate a source of geo-referenced yield data that can enable growers to document the extent of spatial yield variability within fields.
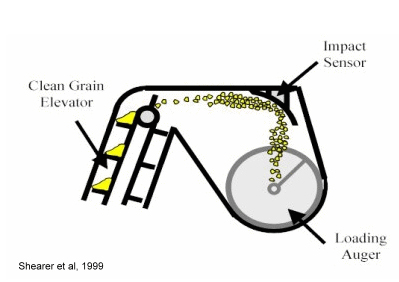
Most yield monitor systems operate on the same general principles. Typically, a grain flow impact sensor is located at the top of the clean grain elevator. Grain flow hits the impact sensor on its way to the loading auger. The impact of the grain flow is translated to electrical signals by the sensor. The electrical signal data are translated to estimates of grain flow rate by the yield monitor’s internal software. If equipped with a DGPS receiver, the yield monitor matches the individual yield estimate data points to geographic locations in the field.
Yield estimates on a whole field or individual load basis made by a well-calibrated yield monitor are accurate in the sense that they often very closely match yield estimates calculated from weigh wagons or commercial weigh scales. However, to achieve a satisfactory level of accuracy, yield monitors must be “trained” to correctly interpret the electrical signals generated by the impact sensor into estimates of grain flow rate . Some background information may help you better understand the nature of and importance of faithfully and regularly calibrating yield monitors.
Calibrating a yield monitor simply requires the harvest of individual “loads” of grain that represent a range of grain flow rates (i.e., a range of yield levels) expected in the field(s) to be harvested.The amount of grain required for each calibration “load” ranges from 3,000 to 6,000 lbs (50 to 100 bu grain) depending on the manufacturer’s recommendations for the specific model/make of yield monitor. The grain weight of each “load” is estimated by the yield monitor as the grain is harvested. The grain for that specific “load” is then offloaded from the combine hopper and weighed on weigh wagon or commercial scales. The actual weight is then entered into the yield monitor console and the yield monitor firmware makes adjustments to curve.
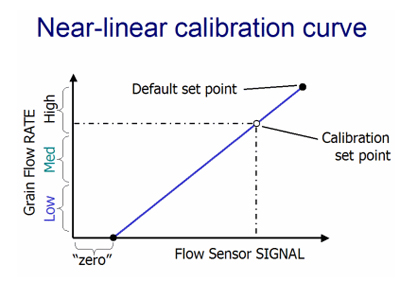
Conceptually, the calibration process is about fitting a response curve between grain flow rate and flow sensor signal strength in order to estimate low, medium, and high yields. Makes of monitors appear to differ in the nature of the calibration curve that is determined.
Some manufacturers suggest that only one grain load is necessary to perform an accurate calibration. The need for only one grain load implies that the calibration response curve is a straight-line or near-linear relationship between grain flow rates and flow sensor signals. While the standard recommendation is for only one grain load, the “fine print” in the owners’ manual suggests that additional calibration loads may be added to fine-tune the accuracy when necessary.
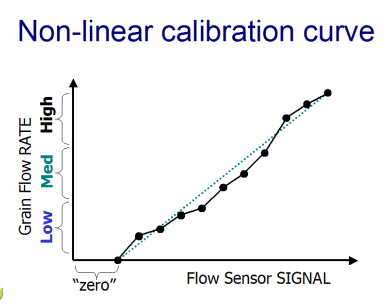
Other manufacturers recommend between 3 and 6 grain loads to perform a satisfactory calibration of the yield monitor. This suggests that the calibration response curve for these yield monitors is not a straight-line, but is rather some sort of non-linear response curve that requires a number of calibration points to best “train” the yield monitor how to interpret the flow sensor signals.
The goal here is to “capture” the full range of grain flow rates (aka yield levels) you expect to encounter during the harvest of your fields. Capturing a range of grain flow rates can be a nuisance because it typically requires harvesting individual full header width “loads” at different speeds or partial header width “loads” at a constant speed. This headache plus the time it takes to off-load and weigh the individual grain loads are among the most common reasons why growers do not faithfully calibrate their yield monitors.
Yield monitor accuracy can be excellent if well-calibrated. Yield estimates by calibrated yield monitors that I use in my field-scale research trials are typically within 1 % of the actual grain weight measured with a weigh wagon or farm scales. Conversely, yield estimates can be very poor if yield monitors are not well-calibrated. The error in accuracy can be as much as 100 % if the yield monitor is taken “off the shelf” and put into service without any calibration. Errors in accuracy can easily range as high as 7 to 10 % late in harvest season if the yield monitor was calibrated only at the beginning of the harvest season. Errors in yield estimates are especially likely if the full anticipated range of harvested grain flow rates are not included in the calibration of the yield monitor.
Well, you may ask... who cares whether or not your yield monitor is providing you with accurate yield estimates. After all, growers are typically paid at the point of sale according to the weights printed on the scale ticket and not according to a yield map. Quite honestly, it also may not matter for simple farm record-keeping purposes.
However, if you want to USE the information that an accurate yield dataset provides, then you should strive to ensure accuracy in the yield estimates made by your yield monitor. Common uses for yield monitor data include comparisons of one field to another, one specific spot in a field to another, one hybrid’s performance to another, early versus late harvest season, and experimental treatments in on-farm field trials.
Yield monitor calibration accuracy can be influenced by yield levels outside the range of grain flow rates used for the yield monitor calibration, by seasonal changes in temperature, by grain moisture content early in the season versus late in the season, by hybrids in terms of their differences for grain weight, grain shape, and grain moisture, and by field topography. Calibrating your yield monitor once a season will typically not be satisfactory. Check the accuracy of the yield monitor calibration occasionally by harvesting and weighing additional calibration loads. Recalibrate the yield monitor when necessary to maintain an acceptable accuracy.
Don’t forget to...
• Also calibrate the combine’s grain moisture sensor.
• Also calibrate for the zero-flow combine vibration.
• Also calibrate the temperature sensor at the beginning of the season.
• Re-read the yield monitor operations manual prior to the harvest season.
• Create a pre-season and in-season yield monitor checklist of all adjustments and settings.
• Go through the yield monitor checklist every morning before beginning the day’s harvest.
Bottom Line
Yield data can be very useful for identifying and diagnosing yield influencing factors in corn or soybean. Yield monitors can also be useful for harvesting on-farm research trials. Yield monitor calibration, yield data processing, and yield data “cleaning” are necessary to ensure accurate yield data. Remember the old adage: “Garbage in….Garbage out”.
Related References
AgLeader Technology. 2009. Manuals and Quick Reference Sheets. [online] <http://www.agleader.com/support.php?Page=manuals>. [URL accessed Sep 2009].
Colvin, T.S. and S. Arslan. 1999. Yield Monitor Accuracy. Potash and Phosphate Institute Publ. SSMG-9. [online, URL accessed Sep 2009].
Grisso, Robert, Mark Alley, and Phil McClellan. 2003. Precision Farming Tools: Yield Monitor. Virginia Cooperative Extension Pub 442-502. [On-line]. Available at <http://www.ext.vt.edu/pubs/bse/442-502/442-502.pdf> [URL accessed Sep 2009].
John Deere. 2009. Quick Reference Guides. [online]. <http://stellarsupport.deere.com/en_US/support/QuickReferenceGuides.html>. [URL accessed Sep 2009].
John Deere. 2009. Operator Manuals & Users Guides. [online]. <http://stellarsupport.deere.com/en_US/support/OperatorManual-UserGuide.html>. [URL accessed Sep 2009].
Shearer, S.A., J.P. Fulton, S.G. McNeill, S.F. Higgins, and T.G. Mueller. 1999. Univ. of Kentucky. [On-line]. Available at <http://www.bae.uky.edu/precag/PrecisionAg/Exten_pubs/pa1.pdf>. [URL accessed Sep 2009].
Watermeier, Nathan. 2004. Yield Monitor Calibration Tips—Making The Most From Your Data. Ohio State Univ. Extension Pub ANR-8-04. [On-line]. Available at <http://ohioline.osu.edu/anr-fact/pdf/0008.pdf> [URL accessed Sep 2009].
![]()
Reddish-Purple Corn Plants Late in the Season - (Bob Nielsen)
Seems like every August, about mid-month or later, some curious farmer calls his local agronomist to complain that he is puzzled over the preponderance of puzzling purplish plants proliferating in his production paddocks. Unless you are an Ohio State or a Nebraska football fan, the sudden appearance of reddish plants in your fields late in the season can be a bit disconcerting.
As I’ve indicated in an article about early-season purpling in young corn, purpling of corn plant tissue results from the formation of a reddish-purple anthocyanin pigments that occur in the form of water-soluble cyanidin glucosides or pelargonidin glucosides (Kim, 1998). A hybrid’s genetic makeup greatly determines whether corn plants are able to produce anthocyanin. A hybrid may have none, one, or many genes that can trigger production of anthocyanin. Purpling can also appear in the silks, anthers and even coleoptile tips of a corn plant.
Most researchers agree that these pigments develop early in the season in young plants in direct response to a number of stresses that limit the plants’ ability to fully utilize the photosynthates produced during the day. These stresses include cool night temperatures, root restrictions, and water stress (both waterlogged and droughty conditions). Since the anthocyanin occurs in the form of a sugar-containing glucoside, the availability of high concentrations of sugar in the leaves (photosynthesis during bright, sunny days) further encourages the pigment formation. If fields are stressed by other factors such as soil compaction, herbicide injury, disease damage, or insect injury, the purpling becomes even more pronounced.
Late in the season, the explanation for the production of these reddish-purple pigments is fundamentally the same (an abundance of photosynthetic sugars in the stalk and leaves) though the fundamental cause of the sugar buildup is different. Late in the season, stalks and leaves will turn reddish-purple when kernel set on the developing ear is minimal or non-existent (i.e., barren ears). The presence of a partially or totally barren ear represents a tremendous reduction in the consumption of photosynthates being generated by that great, big solar-powered photosynthetic factory we call a corn field. Thus, puzzling purplish pigmented plants can develop in response to poor pollination success, severe kernel abortion, severe silk-balling, or arrested ears of various shapes and sizes.
While purpling is not the cause of the problem, it’s existence certainly represents the proverbial “red flag” that signals serious problems with ear size or kernel set.
Related References
Dixon, Richard A. and Nancy L. Paiva. 1995. Stress-lnduced Phenylpropanoid Metabolism. The Plant Cell 7:1085-1097. American Society of Plant Physiologists. [On-Line]. Available at <http://www.plantcell.org/cgi/reprint/7/7/1085>. [URL accessed Sep 2009].
Kim, Jae Hak. 1998. Maize Anthocyanin Pathway. Pennsylvania State Univ. [On-Line]. Available at <http://scripts.cac.psu.edu/courses/plphy/plphy597_hef1/mpath.html>. [URL accessed Sep 2009]. Editorial note: This link is for biochemistry fans!
Nielsen, R.L. (Bob). 2008. Kernel Set Scuttlebutt. Corny News Network, Purdue Univ. [online]. <http://www.kingcorn.org/news/timeless/KernelSet.html> [URL accessed Sep 2009].
Nielsen, R.L. (Bob). 2009. Blunt Ear Syndrome....Again. Corny News Network, Purdue Univ. [online]. <http://www.kingcorn.org/news/timeless/BluntEars.html> [URL accessed Sep 2009].
Nielsen, R.L. (Bob). 2009. Prevalent Purple Plants Possibly Puzzle Producers. Corny News Network, Purdue Univ. [online]. <http://www.kingcorn.org/news/timeless/PurpleCorn.html> [URL accessed Sep 2009].
![]()
Corn Ear Damage Caused by Bird Feeding - (Bob Nielsen)
Blackbirds, grackles, and crows often feed on developing ears in corn fields following pollination early in the grain filling period. They seem particularly attracted to developing kernels near the so-called milk stage or “roasting ear” stage.
Large flocks of these birds can cause a tremendous amount of damage and will often target one hybrid over another. Damage is often most prevalent along field edges and nearby wooded areas, but can extend throughout a large field.
The symptoms that immediately catch the eye are the missing or damaged kernels on the cobs. Damaged cobs often turn brown or black once molds begin infecting the damaged tissue. The evidence that clearly identifies birds as the culprits are the shredded husk leaves that result from the birds working their way to the kernels.
It is not uncommon for birds to be attracted to only one of several hybrids in a field or to one field over another adjacent one. The reasons for this are not clear, but probably related to slight differences in kernel maturity or other kernel characteristics between hybrids. Bollinger and Caslick (1985) indicated that kernel maturity (as measured by date of silking) was the most important factor in determining the level of blackbird damage to corn. The second most important factor was the peak population level of corn rootworm beetles. Within a field, the degree to which the husk leaves extended beyond the tip of the ears was strongly correlated with the severity of damage. In an earlier paper, the same two authors (1984) indicated that as little as an average 10% ear damage throughout a field was associated with an estimated 5,000 birds. The thought of so many birds in one field brings back bad childhood memories of watching Alfred Hitchcock’s classic 1963 horror movie “The Birds”!
Such bird feeding damage predisposes the ears to the development of various ear molds and rots, some of which may subsequently lead to the development of dangerous mycotoxins (Voight, 2008; Woloshuk, 2001). If you are concerned about the potential for mycotoxins from moldy grain on bird-damaged ears, you can send grain samples to Purdue’s Animal Disease Diagnostic Laboratory (ADDL) for a mycotoxin screen. The submission form can be downloaded from the ADDL Web site at <http://www.addl.purdue.edu/Submit/Forms.htm>. Call the ADDL at 765-494-7440 for details on sample submission and cost for the screening.
Related References
Bollinger, Eric and James Caslick. 1984. Relationships between Bird-Dropping Counts, Corn Damage, and Red-Winged Blackbird Activity in Field Corn. J. Wildlife Mgmt 48(1):209-211.
Bollinger, Eric and James Caslick. 1985. Factors Influencing Blackbird Damage to Field Corn. J. Wildlife Mgmt 49(4):1109-1115.
Voight, Del. 2008. Corn Ear Molds. Penn State Field Crop News, Pennsylvania State Univ. [online] <http://fcn.agronomy.psu.edu/2008/fcn0831.cfm#e> [URL accessed Sep 2009].
Woloshuk, Charles. 2001. Mycotoxins and Mycotoxin Test Kits. Purdue Univ. Extension Publ. BP-47. [online] <http://www.ces.purdue.edu/extmedia/BP/BP-47.html> [URL accessed Sep 2009].