USDA-NIFA Extension IPM Grant
Pest & Crop Newsletter, Entomology Extension, Purdue University
Innocent Insects Feeding on Dead/Dying Seedlings Often Falsely Accused! (Christian Krupke and John Obermeyer)
• Numerous pests and non-pests are being found near poor emerging/slow-growing plants.
• Many are attracted by and feeding on dead/decaying plant material.
As pest managers and producers inspect crops that have been “popping” out of the ground, a multitude of insects and other arthropods are being found in the soil. Some are known pests and several are being falsely accused of causing stand reductions. Here are a few of the more common inhabitants found in and around damaged/rotten seed.
Millipedes are very common, usually dark grey or black arthropods (like insects, they belong to the Phylum Arthropoda-means “jointed foot”), having two pairs of legs per body segment. Milli = thousand, pede = feet. They don’t have that many feet, usually under a hundred or so in local species. They become more prevalent as crop/cover residues becomes more abundant. They are often found in large numbers, but are rarely a pest. This is because they typically feed as scavengers, feeding on dead or decaying materials often associated with seedling blights. They are commonly found near any rotting plant material with other arthropod “recyclers”. Several pest managers have reported numerous millipedes in and around corn kernels/sprouts that have been in the ground for two or more weeks. These kernels were probably the victims of pathogens of some kind (bacteria/fungi) and opportunistic millipedes were merely acting as the “clean-up crew” and hollowing out kernels that were in early stages of decay. A healthy and growing corn plant is of no interest to these organisms.
Immature millipede next to rotting corn seedling
Juvenile (“baby”) earthworms (Lumbricids) and potworms (Enchytrids) are closely related and common animals found in soils. They are small, generally colorless/transparent or off-white, and often less than 1/2 inch long. These worms feed on damaged and decaying plant remains, not live tissue. Therefore they are closely associated with the decaying plant parts and surrounding soil and sometimes wrongly accused of damaging seedlings – in fact, they usually arrive after the seed is dead and are attracted by gasses like CO2 that are given off by bacterial rots. In fact, their mouthparts are incapable of causing damage to live tissue – they don’t have “teeth” and instead are specialized to suck up partially-liquefied material. Remember, tkeep an open mind when diagnosing field problems. As one submitter confessed, he was so convinced that it was an insect problem and therefore looked for anything moving when he couldn’t find grubs or wireworms.
Potworms next to damaged soybean seedling
Many other critters, e.g., mites, symphylans, and springtails, have been observed on or around rotting seeds/seedlings. They are small, some fast moving, and certainly unfamiliar to most. They are not causing the poor emergence/growth, but taking advantage of weak and dying plants (including dead weeds and crop debris from previous years) in various stages of decay, as well as the hospitable environment supplied by atypically wet soils. A decaying corn seed is a rich source of nutrients in the mostly-barren environment of newly-planted fields. As a result, many different organisms will arrive to for the feast, the vast majority are not pests.
Two species of springtails u nder high magnification
![]()
Click here to see the Black Light Trap Catch Report
![]()
Scout and Identify Pigweed Species Now - (Travis Legleiter and Bill Johnson) -
The majority of corn and soybean acres in the state of Indiana have been planted and are quickly advancing through their growth stages. The time to start scouting fields and identifying weeds for post-emergence herbicide applications is quickly approaching, if not already here for many producers.
At our Palmer amaranth research plots located in Cass County we already have Palmer amaranth plants that have emerged and have quickly reached 2-3 true leaves. For those producers living in the Northwestern quadrant of the state, you need to be on high alert for the presence of this weed in your fields. Even if it was not present in your fields last year, you need to be aware of this weed and able to distinguish it from the other pigweed species. Palmer amaranth is potentially the most aggressive agronomic weed Indiana producers have ever dealt with, and must be managed with an aggressive control program. Seed bank populations will increase quickly in fields where Palmer amaranth is not correctly identified or managed leading to several years of expensive control programs to manage this aggressive weed.
The first key, as eluded to earlier, is the correct identification of palmer amaranth and it’s very close amaranth relatives: common waterhemp, redroot pigweed, and smooth pigweed. A large majority of the populations discovered last fall had been misidentified as waterhemp for a number of years, which allowed the populations to spread quickly to unmanageable levels.
Differentiating a redroot or smooth pigweed from Palmer amaranth is as simple as looking for fine hairs on the stems and leaves. Redroot and smooth pigweed will have obvious fine hairs on the stems and leaf surfaces, whereas Palmer amaranth will be hairless.
Identifying the differences between Palmer amaranth and common waterhemp (both species are hairless) is much more difficult, especially at a young age. At the cotyledon and first true leaf stage it is even difficult for trained weed scientist to differentiate Palmer amaranth from common waterhemp. A few differences that you can note as these plants begin to put on the second and third true leaves are as follows. We have also included a few images for your reference.
•Singular hair in the leaf tip notch: a characteristic of all pigweed (amaranth) species is a notch in the leaf tip. In the plants we have examined from around the state of Indiana it has been consistent that the first, second, and/or third true leaf of Palmer amaranth can have a small singular hair that protrudes from the leaf tip notch. This singular hair often does not occur on all leaves, but is likely to occur on at least one leaf in the early growth stages. We have not observed this singular hair in the leaf tip notches of common waterhemp seedling from the state of Indiana.
•Petiole length of the first true leaves: As noted in our Palmer amaranth biology, identification and management publication (WS-51), one of the main characteristic differences in waterhemp and Palmer amaranth is the length of the petioles. In Palmer amaranth plants the petioles can be as long or longer that the leaf blade itself. In the Palmer publication we included pictures of mature plants, but this characteristic can be observed in two to four true leaf plants as well. As Palmer plants begin to add the second and third nodes the first true leaf will begin to rapidly elongate its petiole to capture sunlight outside of the shadow of the newly emerging leaves above it. The petioles of common waterhemp will consistently stay short, and the leaf blades themselves will be elongated to capture more sunlight. The elongating petioles of Palmer amaranth seedlings will also begin give the seedling the characteristic rosette pattern as you look down at the growing point.
These two characteristics are the most evident differences between Palmer amaranth and common waterhemp at the seedling growth stages. As the plants become larger, all of the characteristics noted in our Palmer amaranth publication (WS-51) will become more noticeable and evident. Although proper identification of seedling plants is necessary in order to make herbicide applications at the 3-6” weed heights that has are recommended for post emergence Palmer amaranth control.
More in-depth identification and management information is contained in our Palmer amaranth publication (WS-51) that is available from the Purdue Education Store <https://mdc.itap.purdue.edu/item.asp?itemID=21061#.UaUcH-ujTq0>.
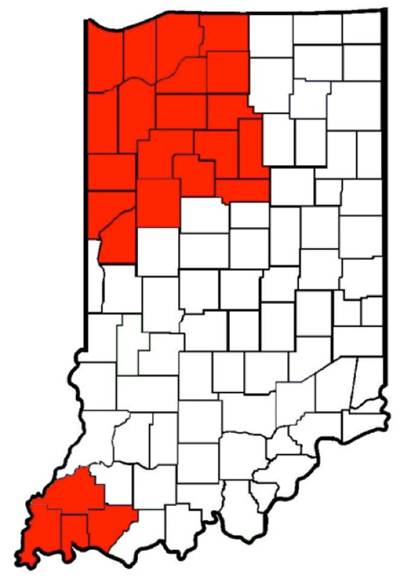
A photo of Palmer amaranth seedlings taken in Cass County Indiana on May 20, 2013
Producers in the hihglighted counties shold be on high alert for the presene of Palmer amaranth in their corn and soybean fields. The highlighted counties have either had Palmer amaranth reported within that county or a neighboring county.
A redroot pigweed or smooth pigweed seedling with fine hairs present on the stem and leaf surfaces.
A common waterhemp seedling lacking the singular hair in the leaf tip notches. Also note the long linear leaf blades attached to short petioles, especially the first true leaves.
A Palmer amaranth seedling with the singular hair in the leaf tip notch of the second true leaf.
Another Palmer amaranth seedling exhibiting the singular leaf tip notch hair on the second true leaf.
A Palmer amaranth seedling at the three true leaf stage that exhibits the elongated petioles of the first true leaves.
Palmer amaranth seedling with the elongated petioles of the first true leaves.
Palmer amaranth seedling with the elongated petioles of the first true leaves.
VIDEO: Palmer Amaranth Identification
Visit our YouTube video on Palmer amaranth identification at: <http://www.youtube.com/watch?v=wNgRvvnPQJ8> .
![]()
Consider Injury from Pop-up Fertilizer When Trouble-shooting Corn – (Jim Camberato) –
Recent submissions to the Purdue University Plant and Pest Diagnostic Laboratory (PPDL) highlight the need for careful consideration of fertilizer rate and source when using pop-up fertilizer.
Long-standing recommendations have suggested an upper limit on the quantity of nitrogen (N) and potassium (as K2O) added as a pop-up fertilizer and the avoidance of certain N fertilizers.
Limits on nutrient quantity in pop-up fertilizer applications are primarily based on the potential for salt injury. When fertilizers dissolve they form ions that compete with the germinating seed and seedling for water. Higher amounts of fertilizer or lower levels of soil moisture increase the concentration of salts in the soil and reduce water availability to the plant.
All fertilizers, even those labeled “low salt”, have the potential to injure germination and seedling establishment when placed with the seed.
Therefore, no more than 5 pounds of N plus K2O per acre are recommended for sandy soils (CEC less than 7) and no more than 8 pounds of N plus K2O are recommended for heavier-textured soils (CEC greater than 7).
Specific N fertilizers, such as urea (including urea-ammonium-nitrate or UAN liquids) and ammonium thiosulfate (ATS), are risky when used as pop-up fertilizers and should be avoided because of their potential to form ammonia and nitrite, which are both toxic to germinating seeds and seedlings.
Fig. 1 Damaged roots and seeds likely from pop-up fertilizer application. More than 14 pounds of N per acre were applied in-furrow, mostly as urea-ammonium nitrate. (Photo courtesy of PPDL).
Root Development in Young Corn – (Bob Nielsen) –
Successful emergence (fast & uniform) does not guarantee successful stand establishment in corn. The next crucial phase is the establishment of a vigorous nodal root system. Success is largely dependent on the initial development of nodal roots from roughly V2 (two leaves with visible leaf collars) to V6.
Corn is a grass and has a fibrous type root system, as compared to soybeans or alfalfa that have tap root systems. Stunting or restriction of the nodal root system during their initial development (e.g., from dry soil, wet soil, cold soil, insect damage, herbicide damage, sidewall compaction, tillage compaction) can easily stunt the entire plant’s development. In fact, when you are attempting to diagnose the cause of stunted corn early in the season, the first place to begin searching for the culprit is below ground.
To better understand rooting development and problems associated with root restrictions, it is important to recognize that root development in corn occurs in two phases. The first phase is the development of the seminal or seed root system. The second phase is the development of the nodal or crown root system.
Corny Trivia: Sometimes you may hear the seminal root system referred to as the primary root system and the nodal root system as the secondary root system. This classification was described by Cannon (1949) and certainly makes chronological sense but always confuses me from the standpoint of importance to the plant.
The Seminal (Seed) Root System
Seminal (seed) roots originate from the scutellar node located within the seed embryo and are composed of the radicle and lateral seminal roots. Even though the seminal roots technically are nodal roots, they are traditionally discussed separately from the nodal roots that develop later from the crown area of the seedling.
The radicle root emerges first from near the tip end of the kernel (Fig. 1) and initially elongate in that direction. The lateral seminal roots emerge later from behind the coleoptile and initially elongate in the opposite direction of the radicle root. However, soon both sets of seminal roots begin to elongate downward in response to gravity (Fig. 3).
Fig. 1. Radicle root and coleoptile of pre-VE seedling.
Fig. 3. Seminal root system of VE seedling, but no evidence yet of nodal root system.
The seminal root system helps sustain seedling development by virtue of water uptake from the soil, but a young corn seedling depends primarily on the energy reserves of the kernel’s starchy endosperm for nourishment until the nodal root system develops later. Once a seedling has emerged (growth stage VE), the rate of new growth of the seminal root systems slows down dramatically as the nodal root system begins to develop from nodes above the mesocotyl.
Even though the seminal root system contributes little to the season-long maintenance of the corn plant, early damage to the radicle or lateral seminal roots can stunt initial seedling development and delay emergence. Such damage will not necessarily cause immediate death of the seedling as long as the kernel itself and mesocotyl remain healthy, but may result in delayed emergence (Fig’s. 12 - 16) or the seedling leafing out underground. As more and more nodal roots become established over time, damage to the seminal root system will have less and less impact on seedling survival.
Fig. 5. First set of nodal roots elongating from lower most node of crown area of V1 corn seedling
Examples of seminal root damage include imbibitional chilling injury (Nielsen, 2012), post-germination injury from lethal or sub-lethal cold temperatures (Nielsen, 2012), and “salt” injury from excessive rates of starter fertilizer placed too close to the kernel. Symptoms of such root damage include retarded root elongation, brown tissue discoloration, prolific root branching, and outright death of root tissue. If the radicle root is damaged severely during its emergence from the kernel, the entire radicle root may die. Once the radicle has elongated a half-inch or so, damage to the root tip will not necessarily kill the entire root, but rather axillary root meristems may initiate extensive root branching in response to damage to the apical meristem.
The Nodal Root System
Nodal roots develop sequentially from individual nodes above the mesocotyl, beginning with the lowermost node in the area of the young seedling known as the “crown”. Once a seedling has reached the V1 stage of development, one can usually identify the first set of nodal roots beginning to elongate from the lowermost node. By the V2 stage of development, the first set of nodal roots are clearly visible and the second set of nodal roots may be starting to elongate from the second node of the seedling. Each set or “whorl” of nodal roots begins to elongate from their respective nodes at about the same timing that each leaf collar emerges from the true whorl of the seedling.
Regarding Seeding Depth & Rooting Depth: Some folks believe that planting corn deeper encourages deeper rooting and vice versa. This belief is mostly myth with a slight hint of truth mixed in. It certainly is true that the depth of the seminal root system is influenced by seeding depth. However, the nodal root system that develops from the crown of the plant is not influenced much at all by seeding depth. This is because the depth of the crown is fairly constant regardless of seeding depth. During emergence of the seedling, the mesocotyl elongates and elevates the coleoptile and crown towards the soil surface. As the coleoptile nears the soil surface, changes in the ratio of red to far red wavelengths of light causes a change in the supply of one or more growth hormones from the coleoptile to the mesocotyl tissue and mesocotyl elongation consequently comes to a halt (Vanderhoef & Briggs, 1978). Since the depth at which the emerging seedling senses the change in red to far red light is fairly constant, the resulting depth of the crown (base) of the coleoptile is nearly the same (1/2 to 3/4 inch) for seeding depths of one inch or greater.
Elongation of the stalk tissue begins between leaf stages V4 and V5. Elongation of the internode above the fifth node usually elevates the sixth node above ground. Subsequent elongation of higher-numbered stalk internodes will result in higher and higher placement of the remaining stalk nodes. Sets of nodal roots that form at above ground stalk nodes are commonly referred to as “brace” roots, but function identically to those nodal roots that form below ground. If surface soil conditions are favorable (moist and not excessively hot), brace roots will successfully penetrate the soil, proliferate, and effectively scavenge the upper soil layers for water and nutrients.
Fig. 7. Seminal and nodal roots of V2 seedling.
Corny Trivia:Root hairs are lateral extensions of root epidermal cells, grow to a length of several millimeters, and number about 200 per sq. millimeter (Gardner et al., 1985). Their typical life span is only about 2 days at moderate temperatures and less so at higher temperatures (Gardner et al., 1985). Root hairs are visible even on the radicle root of a young seedling. Collectively, the surface area represented by root hairs is very large and can account for a large share of nutrient and moisture uptake by the plant.
Corny Trivia: The primary meristem of a root is located near the root tip. Elongation of cells behind the meristem leads to elongation of the root.
A split stalk of an older plant will reveal a “woody” or “pithy” triangle of stalk tissue at the bottom of the corn stalk. This triangle is typically comprised of four stalk nodes, stacked sequentially with #1 at the bottom, whose associated internodes do not elongate. The first internode to elongate is the one above the fourth node, which elongates about 1/4 to 1/2 inch, above which is found the fifth node (usually still below or just at the soil surface). Consequently, five sets or whorls of nodal roots will usually be detectable below ground (one set for each of the below ground stalk nodes).
Fig. 8. Root hairs on a V2 corn seedling.
Corn seedlings transition from nutritional dependence on kernel reserves to nutritional dependence on the nodal roots around the V3 leaf stage. Damage or stress to the first few sets of developing nodal roots during the time period V1 to V5 can severely stunt or delay a corn plant’s development. Damage to the first few sets of nodal roots forces the young seedling to continue its dependence on kernel reserves longer than is optimum. If the kernel reserves are nearly exhausted, continued seedling development is easily stunted and seedling death is not uncommon. Typical stresses that can stunt initial nodal development include fertilizer salt injury, seedling diseases, herbicide injury, insect feeding damage, excessively wet or dry soils, soil compaction (tillage or planter).
Starter Fertilizer Note: The success or not of this transition period that occurs around the V3 stage of development greatly influences whether the crop continues to develop strongly and uniformly. It is not uncommon for fields to develop rather uniformly up to about V3 (because of reliance upon kernel reserves) and then “fall apart” beyond the V3 stage if nodal root development has been compromised by “crappy” growing conditions and the transition from kernel reserves to nodal root support fails or is less than successful. It is at this stage that starter fertilizer plays a role in ensuring that the transition period occurs successfully. At about V3, one or more of the nodal roots will tap into a starter fertilizer band placed approximately 2 inches to the side and 2 inches below the seed (the proverbial 2x2 placement). Starter fertilizer placed in this position has the advantages over seed-placed starter fertilizer because a) its position relative to nodal root development is more advantageous and b) higher rates of nitrogen and/or potassium can be used without risk of injury to the seed during germination and emergence.
A somewhat uncommon, but dramatic, stunted root symptom is what is referred to as the “floppy corn” or “rootless corn” phenomenon (Nielsen, 2010b). This problem occurs most commonly as a result of the detrimental effects of excessively dry surface soil near the time of initial nodal root elongation in young (V2 to V4) corn plants. Young nodal roots that emerge from the crown area of the plant will die if their root tips (and associated meristematic areas) desiccate prior to successful root establishment in moist soil. The crown of a young corn plant is typically located only 3/4 inch or so below the soil surface and so is particularly vulnerable to dry upper soil conditions.
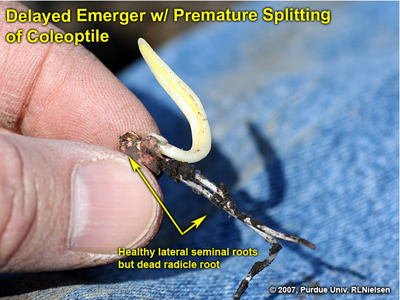
Fig. 12. Delayed emerger with healthy lateral seminal roots but damaged radical root.
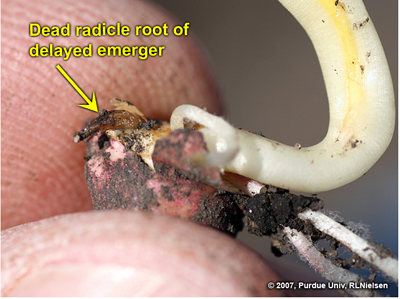
Fig. 14. Dead radicle root on a delayed emerger seedling.
Fig. 15. Delayed emerger with healthy lateral seminal roots and damaged, but alive, radicle root.
Following is an example of a delayed emerger in a field where the “normal” emergers were already at late V1 to early V2. The radicle root was completely destroyed, though the lateral seminal roots were intact and healthy. The coleoptile on this seedling was split down the entire length of its side and would likely result in leafing out underground. The split coleoptile was likely due to the natural continued expansion of the enclosed leaves that would have otherwise emerged normally above ground.
Following is another example of a delayed emerger in the same field where other seedlings were late V1 to early V2. The only visible damage to this delayed emerger was its radicle root whose apical meristem had been injured. The damage was less severe than the previous example and so the seedling was less severely stunted and managed to emerge above ground.
Related References
Cannon, William Austin. 1949. A Tentative Classification of Root Systems. Ecology 30[4], 542-548.
Gardner, Franklin P., R. Brent Pearce, and Roger L. Mitchell. 1985. Physiology of Crop Plants. Iowa State Univ. Press, Ames, IA.
Nielsen, RL (Bob). 2010a. Germination Events in Corn. Corny News Network, Purdue Extension. [On-line] at <http://www.kingcorn.org/news/timeless/GerminationEvents.html>. [URL accessed May 2013].
Nielsen, RL (Bob). 2010b. Rootless or “Floppy” Corn Syndrome. Corny News Network, Purdue Extension. [On-line] at <http://www.kingcorn.org/news/timeless/FloppyCorn.html>. [URL accessed May 2013].
Nielsen, RL (Bob). 2010c. The Emergence Process in Corn. Corny News Network, Purdue Extension. [On-line] at <http://www.kingcorn.org/news/timeless/Emergence.html>. [URL accessed May 2013].
Nielsen, RL (Bob). 2010d. Visual Indicators of Germination in Corn. Corny News Network, Purdue Extension. [On-line] at <http://www.kingcorn.org/news/timeless/GerminationGallery.html>. [URL accessed May 2013].
Nielsen, RL (Bob). 2012. Early-Planted Corn & Cold Weather. Corny News Network, Purdue Extension. [On-line] at <http://www.kingcorn.org/news/articles.12/EarlyCornColdWthr-0412.html>. [URL accessed May 2013].
Vanderhoef, Larry N. and Winslow R. Briggs. 1978. Red Light-inhibited Mesocotyl Elongation in Maize Seedlings. I. The Auxin Hypothesis. Plant Phys. 61:534-537.
![]()
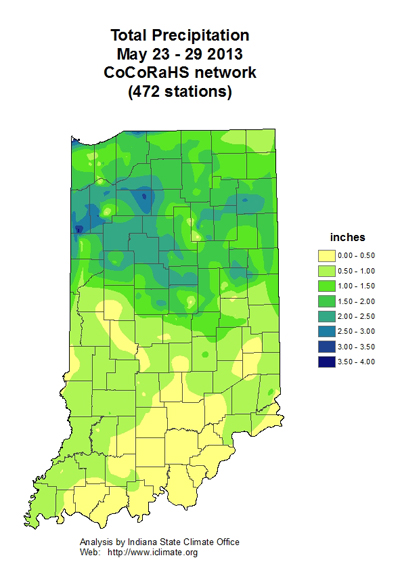
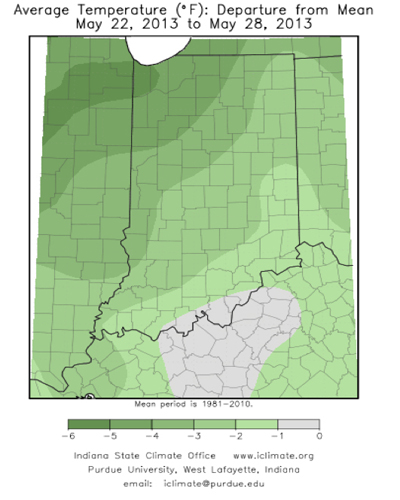
Weather Outlook by NWS OHRFC – (Jim Noel, NOAA/NWS/Ohio River Forecast Center)–
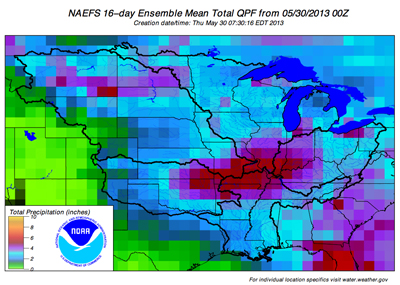
The next two weeks will see slightly above normal temperatures and normal to above normal rainfall. The best chances for rain will be May 30-June 1 and again June 5-7 over the next 2 weeks.
Most places the next 2 weeks will receive 2-4 inches of rainfall with normal being about 2 inches.
The latest rainfall outlook for the next few weeks is available at:
<http://www.erh.noaa.gov/ohrfc/HAS/images/NAEFS16day.pdf>
![]()