- Wheat Virus Diseases Prevalent in Indiana Fields
- Quadris Approved for Use on Greenhouse - Grown Tobacco in Indiana
Nematode Updates- Needle Nematodes on Wheat - (Jamal Faghihi, Christian Krupke, and Virginia Ferris)
We always known that wheat is a good host for the needle nematode and previously warned to be on the lookout for this nematode on wheat. However, we had never isolated needle nematode from wheat roots before last year. In May of 2010 a sample from Jackson County was sent by Mr. Rick Scranton (Pioneer area agronomist), and from this sample we were able to isolate several juveniles of needle nematodes. He reported patches of stunted and abnormal looking wheat. He also indicated that the grower previously had similar issues on corn. Three weeks later, we were unable to recover any needle nematodes from wheat samples in the same field but a large number of needle nematodes were found in adjacent emerging corn samples. The root symptoms are similar to those on corn, but because of the delicate nature of wheat roots, the symptoms are not very obvious. The tips of the roots are truncated and resemble herbicide injury. Like corn, the above ground symptoms do not follow a regular pattern and are usually in patches. We have speculated that needle nematodes start their invasion of wheat roots in the fall. Early in the following spring, when soil temperatures rise to above 50°F, needle nematodes can also invade the established wheat roots. The tiny nematodes aggregate around the roots and with the aid of hollow needle-type mouth parts suck the juice out of the wheat roots. Needle nematode activity usually starts when soil temperatures reach 50°F and usually ceases when soil temperatures rise above 85°F. If you have noticed patches of stunted wheat in sandy soil and previously had similar issues on corn, needle nematode might be the problem. In this case, you may wish to send the entire root system with adjacent soil to the Nematology Laboratory (address below) at Purdue University for analysis, to rule out the nematodes. Samples must be kept cool and prevented from drying. As the soil temperature warms up, this might be a good time to start sampling for needle nematodes on wheat.
Needle nematode damaged area of wheat field (Photo Credit: Rick Scranton)
Severely damaged area of field (Photo Credit: Rick Scranton)
Close-up of needle nematode damaged wheat roots
The sampling procedures for most nematodes are similar. However, samples for the needle nematode must be taken before soil temperatures become too hot, in corn this is usually within 6 weeks after planting. Soil samples must be taken from a depth of 4-6 inches, as close as possible to the infected plants. It is essential to enclose the entire root system with soil surrounding the infected plants. A more detailed sampling procedure can be found on the following website: <http://extension.entm.purdue.edu/nematology/samples.html>.
If you have any questions about corn nematodes or any other kind of plant parasitic nematodes, you can contact Jamal Faghihi at 765-494-5901 or send an email to <jamal@purdue.edu>. Soil samples for nematode analysis can be sent to: Nematology laboratory, Purdue University, Department of Entomology, Smith Hall, 901 W. State Street, West Lafayette, IN 47907-2089. The cost for nematode analysis for each sample remains at $10/sample.
![]()
Black Cutworm Arrival Has Been Detected - (Christian Krupke and John Obermeyer)
• This pest is just beginning its annual invasion to the Midwest.
• Moths arriving in mid to late April pose the greatest threat to crops.
• Heat unit accumulations from dates of intense captures helps plan scouting activity.
Although catches are at barely detectable levels, our pheromone trap cooperators throughout the state have begun monitoring for the black cutworm moth’s arrival into the state from southern Texas and northern Mexico. Warm, moist air currents sweeping up from the Gulf Coast literally lift these moths up into the upper atmosphere and carry them into Midwestern states. The direction that these weather systems move and the number of moths that are carried within them will determine if they are brought into our area and whether or not they will pose a threat to our crops. Last week’s strong winds from the southwest likely brought these early arrivals. More can be expected as storm systems move through the state late this week.
Our black cutworm pheromone trap cooperators will continue to monitor the moth arrival. As we approach the more critical times for moth activity, namely mid to late April, we will be watching for what we refer to as an “intense capture.” This is defined as a period when 9 or more moths are caught in a trap over a two-day period. When and if this occurs, we will begin accumulating heat units (HU base 50°F) to determine when the first cutting of corn by the larvae should occur. This occurs approximately 300 HU after the intense capture. Watch for this information and Black Cutworm Adult Pheromone Trap Reports in future issues of the Pest&Crop.
Black cutworm pheromone trap cooperators are using this bucket trap to monitor their arrival
Wild Garlic and Onion in Wheat – (Bill Johnson and Glenn Nice)
The perennial weeds wild garlic (Allium vineale) and wild onion (Allium canadense) can often be found in wheat fields and occasionally in corn and soybean fields that have been in a no-till system for several years. The reproductive structures, the aerial bulblets, are approximately the same size as wheat grain making them difficult contaminants to remove from harvested wheat. Wheat that is contaminated with these aerial bulblets will be docked for contamination. Although wild onion is a native to North America, wild garlic is a transplant from Europe. It is often wild garlic that is found most often in wheat grain settings, although we occasionally find wild onion as well.
Although wild garlic and wild onion are two different species they look very similar. Both weeds originate from underground bulbs. Their leaves are very similar being basal, long and narrow. However, the two weeds can be differentiated by the fact that wild garlic leaves are nearly round and hollow. Wild onion’s leaves are flat and not hollow (solid). Also the underground bulbs differ; wild garlic’s bulbs have a thin membranous outer coating while wild onion’s bulbs have a fibrous, net-veined coating.
The reason it is important to be able to differentiate will garlic frm wild onion is that control measures for wild onion and wild garlic will differ slightly. Harmony Extra (thifensulfuron + tribenuron) is the most commonly used herbicide to control wild garlic in wheat. Harmony Extra can be applied before planting or after the wheat has reached the 2-leaf stage, but before flag leaf is visible. Apply 0.75 to 0.9 oz/A before the garlic is 12 inches tall and if possible when temperatures are 60 degrees or higher. Dupont recommends for optimum control to apply sequential (or two separate) applications of 0.75 oz/A. The first, early postemergence treatment should be made after the wheat is at the 2-leaf stage. The second treatment is made before the wheat reaches the flag leaf stage. Apply with NIS at 0.25% v/v or COC at 1% v/v. Harmony SG (thifensulfuron) also has activity on wild garlic. Peak (prosulfuron) can provide good control of wild garlic at 0.5 oz/A and be applied before the second node is detectable in stem elongation. However, Peak’s rotation restrictions to soybean are between 10 and 18 months depending where you are in Indiana, ruling out double crop soybeans.
Control measures for wild onion are more limited. Wild onion can be controlled with 2,4-D. Applications of 2,4-D should be after tillering and before jointing and the use rate should be 1 lb ai/A for effective results.
| Recommendations | ||||
| Treatment | Rates | Application Timing | Active Ingredients | Other Weeds Controlled |
| BEST CHOICE: Harmony Extra + surfactant Sequential | 0.75 oz/A followed by 0.75 oz/A | early post followed by late post between 2-leaf to before flag leaf | thifensulfuron + tribenuron | chickweed, deadnettle, henbit, mustards, wild buckwheat, lambsquarter, pigweed, and groundsel |
| Harmony Extra + surfactant | 0.75 to 0.9 oz/A | between 2-leaf to before flag leaf when wild garlic is less than 12 inches | thifensulfuron + tribenuron | |
| Harmony GT + surfactant | 0.75 to 0.9 oz/A | betweem 2-leaf to before flag leaf when wild garlic is less than 12 inches | thifensulfuron | chickweed, mustards, and groundsel |
![]()
The Benefits of Using a Preemergence Herbicide – (Glenn Nice and Bill Johnson)
Early season weed competition can rob emerging crops of water, nutrients and ultimately yield. The use of a preemergence herbicide can benefit in several ways, especially one with some burndown and residual activity. The first benefit is reducing this early season completion. Grasses removed from the field at the 3-inch size contained equal amounts of nitrogen as corn [Link to article: <http://www.ag.purdue.edu/btny/weedscience/Documents/NitrogenAcc08.pdf>]. At the 12-inch size, the grasses accumulated up to 63 lb of N/A. A second benefit is that it provides a wider window for later postemergence herbicide applications. In Roundup Ready and Liberty Link programs, the use of a preemergence herbicide usually provides a more consistent, season-long control and fewer late-season escapes. Finally, some weeds, such as lambsquarter are controlled with most of the available preemergence herbicides but very few postemergence herbicides are effective on large lambsquarter. For more information on the benefits of preemergent herbicides see the following videos and article [link to <http://www.ag.purdue.edu/btny/weedscience/Documents/BenefitsPRE.pdf>].
![]()
![]()
Wheat Virus Diseases Prevalent in Indiana Fields - (Kiersten Wise and Gail Ruhl)
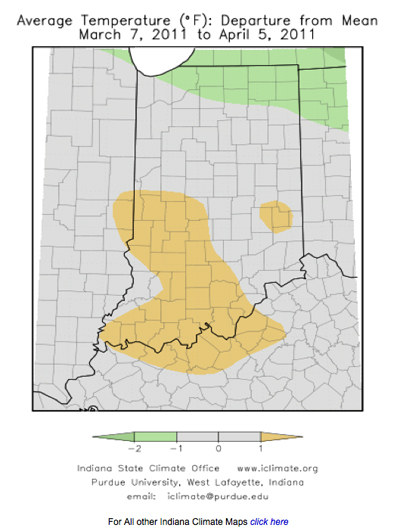
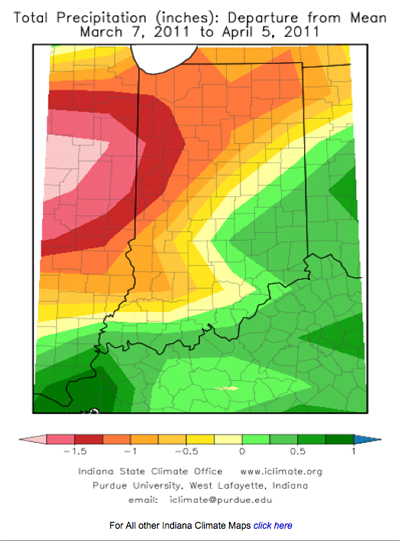
In recent weeks, there have been concerns about patchy yellowing and stunted areas of wheat across fields in Indiana. Scouting reports, and samples submitted to the Purdue Plant and Pest Diagnostic Lab (PPDL) have confirmed the presence of wheat spindle streak (or yellow) mosaic virus (WSSMV), soil-borne wheat mosaic virus (SBWMV), wheat streak mosaic virus (WSMV), and barley yellow dwarf virus (BYDV-RPV strain) in some of these fields.
Virus diseases of wheat are difficult to tell apart in the field and require lab testing for accurate diagnosis. Infected plants typically first appear in uneven patches of yellow or light green within a field, which can be confused with nitrogen deficiency or winter injury. Often, symptoms appear first in low or poorly-drained areas of the field. With soil-borne mosaic viruses (WSSMV and SBWMV), yellow-green streaks may be present on leaves and stunting and/or dieback of leaf tips can occur in infected plants (Figures 1, 2, and 3). The level of symptom expression can depend on variety susceptibility. Soilborne wheat mosaic virus can cause a rosette symptom in susceptible varieties, which results in excessive production of severely stunted tillers. We have also observed reddish coloring on lower leaves of wheat plants infected with soilborne viruses (Figure 4). Plants infected with either virus may produce fewer stems and heads, and have reduced kernel number.
Figure 1. Wheat exhibiting symptoms of soilborne virus diseases (WSSMV; SBWMV)
Figure 2. Yellow "spindels" on leaves characteristic of soilborne virus disease (WSSMV)
Figure 3. Yellow streaks and mosaic on leaves caused by soilborne viruses (WSSMV; WSBMV)
Figure 4. Plants infected with soilborne viruses (WSSMV; SBMWV) may develop reddish-purple coloring on lower leaves
Soilborne wheat mosaic and wheat spindle streak mosaic virus infect wheat plants in the fall. Both viruses are transmitted to wheat roots by the soil-borne fungus Polymyxa graminis. This fungus does not cause damage to wheat by itself, but it infects wheat roots and transmits the viruses to wheat plants. Symptoms of virus infection are not apparent until spring, and the severity of symptom expression depends on variety susceptibility and weather. Prolonged cool temperatures in spring (under 60°F) enhance symptom development of both diseases in infected fields. Often, warmer temperatures in spring will help reduce the appearance of symptoms and plants will appear to recover. Yield may not be reduced if the symptoms and distribution of virus within a field are limited, but severe and/or widespread infections can cause stunting and yield loss. Fields that are severely affected at this point in time may need to be replanted. Consult Extension specialists to determine if replanting is needed in a particular location.
Although no control methods are available to reduce symptoms in currently infected plants, it is still important to get an accurate diagnosis for management of future wheat plantings. Crop rotation may not prevent infection since the fungus that transmits the virus can survive in the soil for over 5 years. Therefore, the best way to manage these diseases is to plant resistant varieties in areas with a previous history of the diseases. Varieties are available with good resistance to one or both of the mosaic virus diseases. Be sure to check the variety if you have problems with both soilborne virus diseases in a single field, since some varieties are resistant to only one virus.
The PPDL provides testing for the presence of WSSMV, SBWMV, Wheat streak mosaic virus (WSMV) and 5 strains of Barley yellow dwarf virus (BYDV) with a multiplex PCR detection assay. Contact the PPDL <http://www.ppdl.purdue.edu/contacts.html> for testing fees. For an accurate diagnosis it is important to dig and submit entire plants exhibiting symptoms (see submission information at <http://www.ppdl.purdue.edu/physical>.
![]()
Quadris Approved for Use on Greenhouse-Grown Tobacco in Indiana - (Kiersten Wise)
Indiana tobacco producers now have another option to control the disease target spot in greenhouse-grown tobacco. The fungicide Quadris (azoxystrobin; Syngenta Crop Protection) has received a special local needs label, known as a 24(c) label in Indiana. This label allows ONE application of Quadris at a rate of 6 fl oz/A, or 0.14 fl oz per 1000 ft2 of float beds in a volume of 5 gal/1000 ft2. This application is separate from Quadris applications that occur in the field, and growers will need a copy of the 24(c) label in their possession if a greenhouse application will occur. A greenhouse application WILL count toward the seasonal limit of Quadris allowed on tobacco. For more information on managing target spot of tobacco, please refer to the newsletter article written by Kenny Seebold of the University of Kentucky: <http://extension.entm.purdue.edu/pestcrop/2011/issue2/index.html#quadris>
2011 Popcorn Agri-Chemical Handbook – (Genny Bertalmio, The Popcorn Board)
The 2011 Popcorn Agri-Chemical Handbook is now available at <http://www.popcorn.org/handbook> to ensure everyone in the popcorn industry is informed about products registered for use on popcorn or in popcorn storage facilities. The handbook lists agri-chemicals registered, special use restrictions and the status of a chemical under special review by the Environmental Protection Agency (EPA). The International Maximum Residue Level (MRL) Database, www.mrldatabase.com <http://www.mrldatabase.com/>, includes popcorn and denotes established levels by the US, Codex, EU and 27 countries.
The Popcorn Board urges you to provide the above links to your growers or download, print and distribute the updated version of this critical information to them. Contact Genny Bertalmio, PH: 312-821-0217 or email: <<ul>gbertalmio@smithbucklin.com/</ul>, for further information or if you require a hard copy.
The Popcorn Board accepts voluntary contributions to ensure continued funding of its efforts to provide this important information to the popcorn industry. Checks should be mailed to The Popcorn Board, 8333 Solutions Center, Chicago, IL 60677-8003.