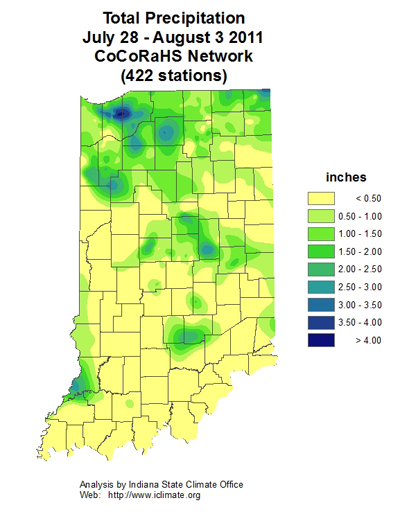
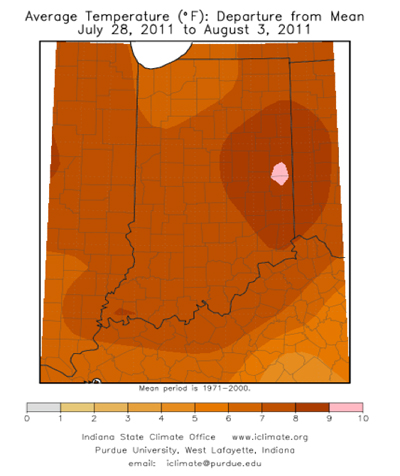
- First In-Field Resistance to Bt Corn Targeting Rootworms Documented in Iowa
- Spider Mites, Rain, and Control
- Western Corn Cutworm Adult Pheromone Trap Report
- Black Light Trap Catch Report
- An Example of "Recovery" from Severe Root-Lodging
- Kernel Set Scuttlebutt
- Effects of Stress During Grain Filling in Corn
First In-Field Resistance to Bt Corn Targeting Rootworms Documented in Iowa – (Christian Krupke)
• Following reports of high damage in Iowa, lab studies revealed resistance to Bt hybrids expressing Cry3Bb1 toxin (found in Monsanto hybrids targeting rootworms).
• Field locations where resistance was documented were characterized by high rootworm pressure, with a history of continuous Bt corn planting.
• This highlights the importance of refuge planting, and Indiana producers should remain vigilant.
Regular readers of Pest&Crop may recall that we have mentioned a few times that corn rootworm Bt toxins are not high dose toxins – meaning that many larvae survive exposure and reach adulthood on each acre of these hybrids. This is one of the reasons that the refuge is so critical in stewardship of this valuable IPM tool. Those points were underscored by the publication last week of the research findings of Iowa State entomologist Aaron Gassman and co-authors. After receiving persistent reports of high damage to Bt corn in northeastern Iowa, the group collected adults and eggs from the area. Rearing the larvae in the laboratory on Bt hybrids revealed that the larvae were able to survive on Bt corn hybrids expressing the Cry3Bb1 toxin at levels similar to survival on non-Bt corn. Hybrids expressing this toxin include those formerly labeled as Yieldgard RW and VT3 hybrids. This toxin is also one of the proteins found in SmartStax hybrids. The good news is that the study tested the other major toxins deployed in North America against this pest, Cry34/35 (found in Herculex hybrids targeting rootworms and also in SmartStax hybrids), and no enhanced survival was found. Although Cry3bb1 and Cry34/35 toxins are different, they are similar enough that cross-resistance (where surviving exposure to one toxin confers some level of survival to another), was a possibility worth investigating. No evidence of cross-resistance was found in these rootworm populations. The next questions to tackle involve untangling the mechanisms behind how these insects are able to survive toxin exposure – what combination of physiological and behavioral traits are at work here? Understanding these mechanisms will undoubtedly help find solutions and plan future control technologies.
Cry3Bb1 traited (left) versus untreated (right) roots from Purdue efficacy trial a few years ago
Given that other researchers have reported that Bt resistance is fairly easy to select for in the lab, we strongly suspected it was just a matter of time before we would see it in the field. The majority of corn planted in the US is Bt corn, and the Cry3bb1 toxin is the major one deployed against rootworms. So what does this mean for other corn-producing areas, including Indiana? It certainly is not good news, but it is not a total disaster either. First off, it demonstrates again what a remarkably adaptable pest the western corn rootworm is. There is no "putting the genie back in the bottle," and resistance in these areas is a problem that won't go away. This is an alert to keep our eyes open for similar occurrences elsewhere – i.e. Indiana. However, the vast majority of Bt corn continues to perform well and be a boon for producers. At the same time, remember that rootworms are the key pest of corn here in Indiana as well, and there is no reason why we could not see resistance occur at some point, either to this or other toxins. The fields to watch are those where the selection pressure upon the pest is highest: namely continuous Bt corn in areas of high rootworm pressure. Some northwestern and north-central Indiana fields may fit this description. In general, however, we have a more diverse cropping system than Iowa with a large proportion of fields rotating corn with soybeans. That helps delay resistance. Planting the recommended refuge certainly helps, and although compliance with the refuge requirements has been falling in recent years, this serves as a stark reminder of how important it is.
Finally, this research also underscores the fact that growers and consultants are not only the stewards of this technology, but they serve as the eyes and ears to report problem fields and initiate further study. None of this research would have been done without reports to Iowa State University extension staff, so please keep your eyes open and don't hesitate to report potential problems.
The full paper can be downloaded and read here:
<doi:10.1371/journal.pone.0022629>.
![]()
Spider Mites, Rain, and Control – (Christian Krupke and John Obermeyer)
• Rainfall has very little direct effect on spider mite populations, although rain and high humidity encourage beneficial fungal pathogens
• Depending on future weather, rain may offer a brief respite to spider mite damage and spread.
• Dimethoate is best to control mites, chlorpyrifos is best option when soybean aphid are also present
Scattered thunderstorms have brought moisture relief to some parched areas of Indiana. Now the question concerning spider mites is whether the rain will "control" them. Before attempting to answer the question, let's review the factors that have combined to create a spider mite problem in soybean fields.
Extended hot and dry conditions will:
1) encourage the movement of spider mites from drying field margins and roadside weeds onto soybean
2) favor rapid (sometimes explosive) reproduction of spider mites
3( cause spider mites to increase their feeding rates, while also allowing greater nutrient uptake from stressed plants. This creates a feedback loop that makes plants more stressed, leading to more mites, etc.
4) dramatically reduce fungal pathogens that normally keep spider mites in check
In contrast, a significant rainfall (1 inch or more) followed by high humidity will:
1) hydrate plants, removing stress and reducing the concentration of nutrients available to mites in the tissues
2) encourage the growth, development, and spread of beneficial fungal pathogens
3) physically kill spider mites by dislodging them from the plant (this is relatively minor)
The bottom line here is that rainfall does not make the spider mites go away, it merely helps plants recover and slows down the reproductive rate of mite populations. Mites are always in each and every field at some level. Unless fungal pathogens wipe them out (given continual high humidity), they will lurk and may possibly return to an outbreak situation should the weather return to hot and dry. Spider mite damage is irreversible. That is, leaves that are severely bronzed or brown will not "green back up" and contribute to the yield of the field.
Two pesticides are recommended to control spider mites. These include dimethoate and chlorpyrifos. Dimethoate is the most efficacious of these compounds for mite control. If soybean aphid is also present in the field, then chlorpyrifos would be a good choice to suppress both pests. Neither of these products will control spider mite eggs, however, and each will provide about 7 to 10 days at most of residual activity. Proper placement of these pesticides is the key to successful control. Nozzle pressures of 40 psi with fine to medium droplet size and 30-40 gallons of water/acre for ground application helps distribute the pesticide throughout the foliage.
Stippling, beginning of spider mite leaf damage
Spider mite bronzed plants near the field edge.
Click here to see the Black Light Trap Catch Report
![]()
Click here to see the Western Bean Cutworm Adult Pheromone Trap Report
![]()
Spraying Fungicides on Drought-Stressed Corn – (Kiersten Wise)
Indiana's "second" corn crop that was planted in late May or early June is at or approaching the tasseling and silking growth stage. In many areas, this late-planted corn is short, uneven in growth stage, and is experiencing nitrogen or other nutrient deficiencies. These problems are compounded by the fact that the tasseling and soon to tassel corn is going through pollination under high temperatures and low soil moisture (Figure 1). Extension corn Agronomists across the Midwest have been discussing the impact that the heat and lack of moisture can have on corn yields: http://graincrops.blogspot.com/2011/07/high-temeperatures-could-hurt-corn.html. The bottom line is that yields may be impacted not only by the late planting date, but also by the current weather conditions. Growers interested in maximizing remaining yield potential may be considering spraying this corn with a fungicide in the hopes that these applications will improve plant water use efficiency, and/or alleviate plant stress through grain fill.
Applying fungicides for purposes other than disease control has not proven to be consistently profitable in replicated field research trials across the Midwest. As we have reported in previous articles, the most likely chance to recoup the investment from a fungicide application is when fungicides are used in response to disease presence or disease risk factors. Across Indiana, development of foliar fungal diseases such as gray leaf spot and northern corn leaf blight has overall been low. Disease development is slowed under high temperatures (above 85-90°F) and dry conditions, and in many fields, lesions of gray leaf spot are well below the ear leaf, or hard to detect (Figure 2). Our scouting thresholds (http://extension.entm.purdue.edu/pestcrop/2011/issue15/index.html) would indicate that in these situations there is not enough disease pressure to warrant a fungicide application.
Additionally, there are very few replicated field research trials that examine the true effects of fungicides on plant water use efficiency and ability to alleviate drought stress. If fungicides will be applied to corn for these purposes, it would be beneficial to work with the applicator and leave several untreated strips within the field that can be harvested and yields averaged to determine if there was an actual yield benefit due to the fungicide application. This does require additional planning and effort, but it can mean the difference between knowing if the fungicides were worth the investment and whether or not they work under your specific production conditions.
Figure 1. Corn showing symptoms of moisture and heat stress
Figure 2. Lower leaves of corn plant with little or no gray leaf spot lesions present (Picture taken in Northeast IN on 8/1/11).
![]()
Ear Rot Review – (Kiersten Wise and Charles Woloshuk)
April-planted corn is in milk or early dough stages, and weather conditions have been favorable for ear rot disease development, especially in southern IN. Three potential ear rot problems for 2011 are discussed in this article.
Aspergillus ear rot is common in hot, dry years and is most prevalent on drought-stressed plants. This ear rot is caused by the fungus Aspergillus flavus, which also produces a mycotoxin known as aflatoxin. Aflatoxin is a very toxic carcinogen, and livestock that consume contaminated grain may be at risk for many health problems. Aspergillus ear rot appear as stunted ears that have an olive-green dusty mold under the husk on plants that are severely affected by drought stress and warm overnight temperatures. (Figure 1). Low levels of Aspergillus ear rot were detected in 2010 in southern IN, and conditions are once again favorable for disease development in 2011. More information about this disease can be found at: http://www.extension.purdue.edu/extmedia/BP/BP-83-W.pdf.
Figure 1. Aspergillus ear rot of corn (Photo by Burt Bluhm)
Fusarium ear rot, caused by Fusarium verticillioides, is also more severe under hot and dry weather conditions. This fungus also produces a mycotoxin known as fumonisin, which is especially toxic to horses and swine. Symptoms of Fusarium include white, pink, or gray kernels scattered across the ear and are often associated with insect feeding damage. In some hybrids, a white streaking can appear in kernels, which is known as a "starburst" pattern.
Figure 2. Fusarium ear rot of corn (Photo Charles Woloshuk)
Diplodia ear rot, caused by the fungus Stenocarpella maydis, is a common ear rot across IN, but usually appears first in southern regions. The fungus infects corn at and during silking, and infection and disease development are favored by wet weather. April planted corn in Indiana experienced wet, humid weather at silking, which may have favored infection in susceptible hybrids. Infected ears often have bleached husks with tiny black specks on the outer layer (Figure 3). Removal of the husk of an infected ear will reveal white fuzzy growth of the pathogen between the kernels, which often starts at the base of the ear (Figure 4). The cob can also appear rotted. More information about this disease can be found at: http://www.extension.purdue.edu/extmedia/BP/BP-75-W.pdf.
Figure 3. Corn husk with symptoms of Diplodia er rot (Photo by Paty Romero)
Figure 4. Ears with white mold at the base of the cob, which is indicative of Diplodia ear rot
At this point in the season, there are no management strategies for reducing ear rots if detected. Producers should scout fields prior to harvest and determine the level of incidence of the disease in the field. If any Aspergillus ear rot is observed in a field, affected areas should be harvested early and grain segregated to avoid aflatoxin contamination of non-infected grain. Fields affected by Diplodia and Fusarium ear rots should also be harvested prior to other non-affected fields. All grain contaminated by any ear rot fungus should be stored separately from good grain, and stored below 15% moisture to prevent further growth of fungi.
![]()
An Example of "Recovery" From Severe Root-Lodging – (Bob Nielsen)
A thunderstorm with strong winds flattened hundreds of acres in eastern Indiana late in the afternoon of 22 July as a result of plants being partially uprooted. The appearance of damaged fields the morning after was demoralizing to growers and casual observers alike.
However, because many of these fields were planted extremely late due to a wet planting season, the plants were still in the late vegetative phase of development (1 to 2 weeks before tasseling) and, most importantly, still in the process of stalk elongation. Root-lodged stalks that are still elongating can respond to such root-lodging by slowly bending or "goose-necking" in an attempt to regain an upright stance. Such "goose-necking" is the result of changes in the distribution of plant growth hormones in the stalk tissue that cause more rapid elongation on the bottom side of the nearly horizontal stalks than on the top side.
As long as root damage caused by the lodging is not extreme and there is adequate soil moisture to foster additional root development during the recovery period, flattened fields of corn at these growth stages can "recover" fairly well. I put the word "recover" in quotes because severely root-lodged fields will usually not recover completely. However, if the damaged plants can goose-neck sufficiently and quickly enough by the time the field moves into the critical tassel/silk pollination period such that the portion of the stalk containing the silked ears is again upright, then pollination will likely be successful.
The three photos accompanying this article are from a 30-acre field at the Davis-Purdue Ag Center in Randolph County. The storm moved through the area late in the afternoon of 22 July and severely flattened hundreds, if not thousands, of corn acres in the area. The field had been planted 3 June, much later than desired because of the frustratingly late, wet planting season. However, the good news was that plant development was consequently delayed relative to the calendar and the field was still about one week away from tasseling and pollination. I say "good news" because the plants were still in their rapid growth phase with stalks still rapidly elongating.
The first photo shows the appearance of the damaged field the morning after the storm. I estimated 80-90% of the plants were root-lodged and nearly flat to the ground. The second photo shows the same field six days late on 28 July and the change in appearance is amazing. Dramatic bending of the horizontal lower stalk tissue resulted in "goose-necked" plants and, more importantly, enough upright growth to place the silking ears in a position to be exposed to pollen from the tassels.
Time will tell to what extent yield will be decreased and, unfortunately, there is no good comparison to even determine how much yield will be lost. With the hope that weather conditions during grain filling improves, the next big challenge will be harvesting the crop because of the difficulty of moving the combine header through the yet root-lodged lower portions of the crop canopy.
Related reading
Nielsen, R.L. (Bob). 2011. Prospects of Recovery for Root-Lodged Corn. Corny News Network, Purdue Univ. Extension. [online] Available at http://www.kingcorn.org/news/articles.11/FlatCorn-0726.html [URL accessed July 2011].
![]()
Kernel Set Scuttlebutt – (Bob Nielsen)
"Scuttlebutt": The cask of drinking water on ships was called a scuttlebutt and since sailors exchanged gossip when they gathered at the scuttlebutt for a drink of water, scuttlebutt became U.S. Navy slang for gossip or rumors. A butt was a wooden cask, which held water or other liquids; to scuttle is to drill a hole, as for tapping a cask.
Nautical Terms and Phrases, NAVAL HISTORICAL CENTER, Washington DC 20374-5060.
Online at http://www.ussbrainedd630.com/terms.htm [URL accessed Aug 2011].
The post-pollination scuttlebutt overheard in coffee shops throughout Indiana during late summer often revolves around the potential for severe stress that might reduce kernel set or kernel size in neighborhood cornfields. Growers' interest in this topic obviously lies with the fact that the number of kernels per ear is a rather important component of total grain yield per acre for corn.
Poor kernel set, meaning an unacceptably low kernel number per ear, is not surprising in fields that are obviously severely stressed by drought, but can also occur in fields that otherwise appear to be in good shape. Good or poor kernel set is determined from pollination through the early stages of kernel development; typically 2 to 3 weeks after pollination is complete.
Problems with kernel set stem from ineffective pollination, kernel abortion, or both. Distinguishing between these two symptoms is easy. Determining the exact cause of the problem is sometimes difficult.
Potential Yield Loss
The potential loss in grain yield caused by lower kernel numbers per ear can be estimated using the formula of the so-called Yield Component Method first described by the Univ. of Illinois many years ago (Univ. of Illinois, 2009; Nielsen, 2010a). For example, the loss of only 1 kernel per row for a hybrid with 16-row ears and a stand count of 30,000 ears per acre would equal a potential yield loss of approximately 5 bushels per acre (1 [kernel] x 16 [rows] x 30 [thousand ears per acre] divided by 90 [thousand kernels per bushel]).
Ineffective Pollination
Poor kernel set may be caused by ineffective pollination and the subsequent failure to fertilize ovules on the cob. Ineffective pollination is characterized by an absence of noticeable kernel development. In other words, all you see is cob tissue. Pollination problems may be due to several stress factors this year, sometimes working together to influence kernel set.
Severe drought stress, aggravated by excessive heat, can delay silk emergence to the extent that pollen shed is complete or nearly complete by the time the silks finally emerge from the husk. Without a pollen source, ovule fertilization cannot occur.
Persistent severe silk clipping by insects such as the corn rootworm beetle or Japanese beetle throughout the active pollen shed period can also limit the success of pollination. The simultaneous effects of severe drought stress on silk emergence can easily amplify the consequences of severe silk clipping.
Severe drought stress coupled with excessive heat and low humidity can sometimes desiccate emerged silks to the point that they are no longer receptive to pollen grain germination. I suspect this is low on the list of possible stressors for Indiana most years (because of our typically high humidity levels), but may play a role in some fields once in a while. Similarly, I doubt that pollen viability is usually an issue for Indiana cornfields because temperatures in the low 90's are usually not great enough to kill pollen.
Consecutive days of persistent rainfall or showers that keep tassels wet for many hours of a day can delay or interfere with anther exsertion and pollen shed. Such a weather period does not typically occur in Indiana, but the remnants of Hurricane Dennis that visited many parts of Indiana in early July of 2005 may have influenced kernel set in some fields that were trying to pollinate during that week as a result of the many days of showery humid weather (coupled with the excessive cloudiness and its effect on photosynthesis).
Exceptionally long potential ears resulting from good weather during ear size determination (not an issue for some Indiana fields this year) sometimes fail to pollinate the final kernels near the tip of the cob. Remember, butt silks emerge first and tip silks emerge last. With oversized ears, sometimes those tip silks emerge after all the pollen has been shed. See the "Word to the Wise" below.
Kernel Abortion
Poor kernel set can also be a reflection of kernel abortion following successful fertilization of the ovules on the cob. In contrast to ineffective pollination, initial kernel development obviously precedes kernel abortion, so the symptoms are usually shriveled remnants of kernels that may be whitish- or yellowish-translucent.
Kernel abortion results from severe stresses that greatly reduce the overall photosynthetic output of the plant very early in the post-pollination grain-filling period. Obvious photosynthetic stressors include severe drought & heat stress, consecutive days of excessively cloudy weather and significant loss of photosynthetically active leaf area (e.g., hail damage, leaf diseases, insect damage, nutrient deficiency).
Warm nights during pollination and early grain fill may indirectly affect survival of developing kernels. Research suggests that the increased rate of kernel development due to warmer temperatures lowers the available amount of photosynthate per unit of thermal time; which then becomes a stressor to kernel development particularly at the tip of the ear, leading to kernel abortion (Cantarero et al., 1999).
Final Food for Thought
A plethora (meaning a whole lot) of blank cob tips can quickly ruin the joy of walking a cornfield in the middle of August. Before getting too bent out of shape over the missing kernels, remember to count the number of harvestable kernels on those ears. Sometimes, ears exhibit 1 to 2 inches of blank tips; yet still contain 16 rows by 30 to 35 harvestable kernels per row. Those are perfectly acceptable ear sizes in a year where dry weather has been a concern.
Related References
Cantarero, M.G., A.G. Cirilo, and F.H. Andrade. 1999. Night temperature at silking affects kernel set in maize. Crop Sci 39:703-710.
Nielsen, R.L. (Bob). 2008. Grain Fill Stages in Corn. Corny News Network, Purdue Univ. [On-Line]. Available at http://www.kingcorn.org/news/timeless/GrainFill.html [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2009. Yield Loss Potential During Grain Fill. Corny News Network, Purdue Univ. [On-Line]. Available at http://www.kingcorn.org/news/timeless/GrainFillStress.html [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2010a. Estimating Corn Grain Yield Prior to Harvest. Corny News Network, Purdue Univ. [On-Line]. Available at http://www.kingcorn.org/news/timeless/YldEstMethod.html [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2010b. Silk Emergence. Corny News Network, Purdue Univ. Available [On-Line]. Available at http://www.kingcorn.org/news/timeless/Silks.html [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2010c. Tassel Emergence & Pollen Shed. Corny News Network, Purdue Univ. Available [On-Line]. Available at http://www.kingcorn.org/news/timeless/Tassels.html [URL accessed Aug 2011].
Univ. of Illinois. 2099. Illinois Agronomy Handbook, 24th ed.. https://pubsplus.illinois.edu/C1394.html [URL accessed Aug 2011].
![]()
Effects of Stress During Grain Filling in Corn – (Bob Nielsen)
Yield loss during grain fill can occur from 1) stand loss, 2) incomplete kernel set, 3) decreased kernel weight, and 4) premature plant death.
Stand Loss During Grain Fill
Yield loss due to stand loss during grain fill is usually greater than that due to stand loss that occurs during the vegetative phase. When stand loss occurs prior to pollination, ear size (number of kernels) on surviving plants may compensate in response to the lesser competition of a thinner stand. Additional compensation may occur during grain fill in terms of greater kernel weight. When stand loss occurs during grain fill, ear size has already been set. Only kernel weight can compensate in response to the lesser competition of a thinner stand.
Incomplete Kernel Set in Corn
Kernel set refers to the degree to which kernels have developed up and down the cob. Incomplete kernel set is not always apparent from "windshield" surveys of a corn field. Husks and cob will continue to lengthen even if kernel set is incomplete. A wonderfully long, robust-looking, healthy green ear shoot can completely mask even a 100 percent failure of pollination or severe kernel abortion.
TECHNICAL TRIVIA:
Pollination is the movement of pollen from the tassels to the silks.
Fertilization is the actual union of the male and female gametes once the pollen tube reaches the ovule.
One of the causes of incomplete kernel set is unsuccessful fertilization of the ovules during pollination. Unsuccessful fertilization results in ovules that never develop into kernels and, subsequently, ears with varying degrees and patterns of incomplete kernel set. Many factors can cause incomplete pollination and distinguishing between them can be very difficult.
Certain insects like corn rootworm beetles and Japanese beetles can interfere with pollination and fertilization by their silk clipping action. These insects feed on pollen and subsequently clip silks as they feed on the pollen that has been captured by the silks. Unusually early or late pollinating fields are often particularly attractive to these insects.
Drought stress may delay silk emergence until pollen shed is nearly or completely finished. During periods of high temperatures, low relative humidities, and inadequate soil moisture levels, exposed silks may also dessicate and become non-receptive to pollen germination.
Unusually favorable conditions prior to pollination that favor ear size determination can result in ears with an unusually high number of potential kernels per row. Remember that silk elongation begins near the butt of the ear and progresses up toward the tip. The tip silks are typically the last to emerge from the husk leaves. If ears are unusually long (many kernels per row), the final silks from the tip of the ear may emerge after all the pollen has been shed.
Occasionally, silk emergence begins to occur several days prior to the anthers beginning to shed pollen. Although silks remain viable to pollen germination for up to 10 days after emergence, they begin to slowly deteriorate (age, desiccation) after 4 to 5 days. It is conceivable that kernel set near the butt or base of the cob may fail under these conditions.
Another cause of incomplete kernel set is abortion of fertilized ovules. Aborted kernels are distinguished from unfertilized ovules in that aborted kernels had actually begun development. Aborted kernels will be shrunken, mostly white, often with the yellow embryo visible; compared to normal plump yellow kernels.
Kernels are most susceptible to abortion during the first 2 weeks following pollination, particularly kernels near the tip of the ear. Tip kernels are generally last to be fertilized, less vigorous than the rest, and are most susceptible to abortion. Once kernels have reached the dough stage of development, further yield losses will occur mainly from reductions in kernel dry weight accumulation.
Severe drought stress that continues into the early stages of kernel development (blister and milk stages) can easily abort developing kernels.
Severe nutrient deficiencies (especially nitrogen) can also abort kernels if enough of the photosynthetic "factory" is damaged.
Extensive loss of green leaf tissue by certain leaf diseases, such as common rust or gray leaf spot, by the time pollination occurs may limit photosynthate production enough to cause kernel abortion.
Extensive loss of green leaf tissue from severe hail damage at or shortly after pollination can limit photosynthate production and cause kernel abortion.
Consecutive days of heavily overcast, cloudy conditions may also reduce photosynthesis enough to cause abortion in recently fertilized ovules.
Decreased Kernel Weight
Severe stress during dough and dent stages of grain fill often results in premature kernel black layer formation and thus decreases grain yield due to decreased kernel size and weight. Decreased kernel weight can result from severe drought and heat stress during grain fill; extensive European corn borer tunneling (especially in the ear shanks); loss of photosynthetic leaf area by hail, insects, or disease early in grain fill; and killing fall frosts prior to normal black layer development.
Once grain has reached physiological maturity, stress will have no further physiological effect on final yield, because final yield is already achieved. Stalk and ear rots, however, can continue to develop after corn has reached physiological maturity and indirectly reduce grain yield as a result of increased mechanical harvest losses.
Premature Plant Death
A killing fall frost prior to physiological maturity can cause premature leaf death or whole plant death. Premature death of leaves results in yield losses because the photosynthetic "factory" output is greatly reduced. The plant may remobilize stored carbohydrates from the leaves or stalk tissue to the developing ears, but yield potential will still be lost. Approximate yield losses due to premature death of leaves but not stalks range from 36, 31, and 7% when the leaf death occurs at R4 (dough), R5 (full dent), and half-milkline stages of kernel development, respectively (Afuakwa & Crookston, 1984).
Premature death of whole plants results in greater yield losses than if only leaves are killed. Death of all plant tissue prevents any further remobilization of stored carbohydrates to the developing ear. Whole plant death that occurs before normal black layer formation will cause premature black layer development, resulting in incomplete grain fill and lightweight, chaffy grain. Grain moisture will be greater than 35%, requiring substantial field drydown before harvest. Approximate yield losses due to premature whole plant death range from 50, 39, and 12% when the whole plant death occurs at R4 (dough), R5 (full dent), and half-milkline stages of kernel development, respectively (Afuakwa & Crookston, 1984).
A common misconception is that kernel black layer formation sometimes does not occur following a frost or other late-season severe stress. Kernel black layer always occurs. Any severe stress that occurs during the grain fill period will cause premature kernel black layer formation and is related to the reduction in or termination of sucrose (photosynthate) availability to the developing kernels (Afuakwa et al., 1984).
Related References
Afuakwa, J. J. and R. K Crookston. 1984. Using the kernel milk line to visually monitor grain maturity in maize. Crop Sci. 24:687-691.
Afuakwa, J.J., R.K. Crookston, and R.J. Jones. 1984. Effect of Temperature and Sucrose Availability on Kernel Black Layer Formation in Maize. Crop Sci. 24:285-288.
Nielsen, R.L. (Bob). 2007. Ear Size Determination in Corn. Corny News Network, Purdue Univ. [online] http://www.kingcorn.org/news/timeless/EarSize.html. [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2008. Grain Fill Stages in Corn. Corny News Network, Purdue Univ. [online] http://www.kingcorn.org/news/timeless/GrainFill.html. [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2010. Silk Emergence. Corny News Network, Purdue Univ. [online] http://www.kingcorn.org/news/timeless/silks.html. [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2010. Tassel Emergence & Pollen Shed. Corny News Network, Purdue Univ. [online] http://www.kingcorn.org/news/timeless/Tassels.html. [URL accessed Aug 2011].
Nielsen, R.L. (Bob). 2011. Kernel Set Scuttlebutt. Corny News Network, Purdue Univ. [online] http://www.kingcorn.org/news/timeless/KernelSet.html. [URL accessed Aug 2011].
![]()