Pest & Crop Newsletter, Entomology Extension, Purdue University
- Time to Scout Smart: High Risk Fields and Potential Pests
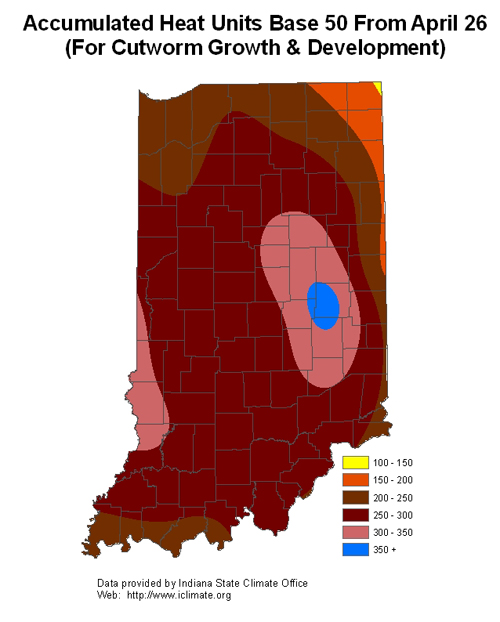
- Accumulated Heat Units Base 50
- Black Light Trap Catch Report
![]()
Time to Scout Smart: High Risk Fields and Potential Pests - (Christian Krupke, John Obermeyer, and Larry Bledsoe)
- Corn planted into grass = armyworm risk
- Wheat in areas of dense growth = armyworm risk
- Corn where weedy growth existed = cutworm risk
- Soybean first emerging is at risk for bean leaf beetle
Corn & Armyworm - Corn that has been no-tilled into an abandoned wheat stand or a grass cover crop should be inspected immediately for armyworm feeding in southern Indiana. Hatched larvae will move from the dying grasses to emerging/emerged corn. Armyworm feeding gives corn a ragged appearance, with feeding extending from the leaf margin toward the midrib. Damage may be so extensive that most of the plant, with the exception of the midrib and stalk, is consumed. A highly damaged plant may recover if the growing point has not been destroyed. If more than 50% of the plants show armyworm feeding and live larvae less than 1-1/4 inches long are numerous in the field, a control may be necessary. Larvae greater than 1-1/4 inches consume a large amount of leaf tissue and are more difficult to control. If armyworm are detected migrating from border areas or waterways within fields, spot treatments in these areas are possible if the problem is identified early enough.

Armyworm corn leaf feeding
Wheat/Grass Pasture & Armyworm - Examine plants in various areas of the field, especially where plant growth is dense. Look for flag leaf feeding, clipped heads, and armyworm droppings on the ground. Shake the plants and count the number of armyworm larvae on the ground and under plant debris. On sunny days, the armyworm will take shelter under crop residue or soil clods. If counts average approximately 5 or more per linear foot of row, the worms are less than 1-1/4 inches long, and leaf feeding is evident, control may be justified. If a significant number of these larvae are present and they are destroying the leaves or the heads, treat immediately.

Armyworm wheat leaf feeding
Corn & Black Cutworm – The moth flight into the state has been rather light this year (remember that weather systems are required to move these moths around), this correlates nicely with the few reports of damage being received. Generally the moths that arrived weeks ago were attracted to winter annual weeds for egg-laying which increases the likelihood of damage from this pest. Mind you…they don’t always read the book! Insecticides, whether soil or seed-applied, should not lull producers into a false sense of security, as larger larvae are able to continue their feeding frenzy. According to black cutworm development models, leaf feeding and plant cutting by larger larvae (1/2” or greater) is possible now throughout most of the state. Timed scouting, and careful assessment of damage can go a long way in preserving a stand of corn.

Cutworm leaf feeding and initial cutting
Soybean & Bean Leaf Beetle – These beetles awoke from their winter’s nap some time ago and have been feeding on forages and other legumes while waiting for soybean to be planted and emerge. Amazingly, they are able to “smell” and subsequently find first emerging soybean, whether near a wood’s edge or in the middle of a large field. Once plants have emerged throughout a field, beetles will typically dissipate to non-economic levels. First planted and/or first emerging soybean seedlings should be inspected for their feeding on cotyledons and unifoliate leaves. Extensive cotyledon damage is cause for serious concern - if cotyledons are being destroyed before the unifoliolate leaves fully emerge or if the growing point is severely damaged, reduced yields are likely. However, once trifoliate leaves have unrolled, soybean can tolerate up to about 40% defoliation without yield loss. It may look ugly, but they can take a beating.

Early bean leaf beetle feeding (Photo credit: Dan Childs)
![]()
Click here to view the
Black Light Trap Catch Report - (John Obermeyer)
Yellow Dwarf of Wheat - (Gregory Shaner and Shawn Conley)
The pronouncement in issue 7 of Pest&Crop about the lack of disease in wheat may have been premature. During the past week, symptoms of yellow dwarf have become evident in many wheat fields. Fields that looked quite good during the jointing and stem elongation phases are now showing the irregular plant height that is one characteristic of this disease. The rather sudden appearance of symptoms in wheat that had looked good may be because the wheat seems shorter than normal this year. It was not until the peduncles elongated that stunting became evident.
Erect flag leaves with purple tips are another diagnostic symptom of yellow dwarf. Overall plant color may be a pale green that suggests nitrogen deficiency. The stunting that results from yellow dwarf is more than cosmetic. Infected plants have a stunted root system as well as short stalks and smaller leaves. Heads are likewise smaller, which reduces yield potential.
Yellow dwarf is caused by any of several closely related species of virus, most of which are grouped into Barley yellow dwarf virus or Cereal yellow dwarf virus. A couple of other species have not yet been formally named. All of these viruses are transmitted only by aphids. The bird cherry-oat aphid (Rhopalosiphum padi), the corn leaf aphid (R. maidis), and the English grain aphid (Sitobion avenae) are common vectors in our area. When a viruliferous aphid feeds on a wheat plant, she transmits the virus to the plant. The aphid does not transmit virus to her progeny. However, shortly after the new little aphids start to feed on an infected plant, they acquire the virus. As they move to healthy plants, they spread the infection.
Infection can occur in fall or spring. Fall infection is more damaging because it results in stunting, reduced tillering, and a predisposition to winter killing. Spring infection causes conspicuous yellowing and purpling of the flag leaf, but not severe stunting. Fields we have looked at were probably infected in the fall.

Field with severe yellow drarf. Note the erect flag leaves with purple tips.
Planting after the fly-free date reduces risk of infection because temperatures are normally low enough after that time that insects are less active. However, last fall daily high temperatures were in the mid 60s into late November, so aphids had plenty of time to reproduce on wheat.

Yellow dwarf can greatly reduce head size. Left: Head from a plant with severe yellow dwarf symptoms; Right: Head from a plant with no symptoms.
What should be done with fields that have yellow dwarf this year? The decision whether to destroy a field and plant soybean or corn depends on how much the wheat yield is likely to be reduced, how quickly the new crop can be planted, and plant back restrictions for any herbicides that may have been applied to the wheat. Estimating yield reduction from yellow dwarf is tricky. To estimate the number of heads per square foot, count heads in several arbitrarily selected 19-in. lengths of row for wheat planted at a row spacing of 7.5 inches. For a high yield, the population of heads should range from 60 to 70 plants/ft2; for an adequate yield, 55 heads/ft2. These densities assume each head has 16 to 18 spikelets and produces 35 kernels. Yellow dwarf reduces head size and spikelet number, so this must be factored into a yield estimate. Another variable, which cannot be estimated before harvest, is kernel size. Yellow dwarf reduces kernel size. In a study conducted in Missouri (Conley et. al.), thousand-kernel weight was reduced from 5 to 10% by yellow dwarf.
We looked at a field with severe yellow dwarf in Tippecanoe County. Heads on healthier plants were at mid anthesis; heads on plants with severe yellow dwarf were not yet flowering or still in the boot. Among 5 samples, the number of heads per square foot ranged from 26 to 54 (average = 42.4), well below the target for an adequate yield. In an arbitrary sample of 20 heads, spikelet numbers ranged from 11 to 18 (average = 13.6). The grower said he customarily got 82 to 90 bu/A of wheat. Using the average stand and spikelet number, we calculated a yield of 64 bu/A, assuming seed will be of normal size. If seed size is reduced, which is likely, the yield will be less.
When a field with yellow dwarf is harvested, the combine header needs to be set lower, to pick up heads on short stems. Combine speed should be reduced to handle the extra straw, and air speed should be reduced to avoid blowing small kernels out with the chaff.
Where yellow dwarf is a more chronic problem, generally south of Indiana, treatment of seed with an insecticide (Cruiser or Gaucho) will reduce buildup of aphids in the fall and therefore reduce the spread of yellow dwarf. In Indiana, yellow dwarf is a sporadic problem, so routine use of an insecticide seed treatment may not be economically justified. Scouting and fall applications of foliar insecticide may prove beneficial when warm weather persists through November and into December. Conley’s research from Missouri indicates a fall aphid threshold of 5 per foot of row and a spring threshold of 50 aphids per foot of row. No variety is highly resistant to yellow dwarf, but some varieties have a useful degree of partial resistance or tolerance.
![]()
Soybean Rust Update - (Greg Shaner)
Plant pathologists in Louisiana reported finding soybean rust on kudzu in Iberia Parish, in southern Louisiana. They found the rust on May 8, but waited for confirmation of their visual diagnosis by molecular tests before making the announcement on May 11. This finding is 53 days earlier than the first detection of rust in Louisiana last year. Although Florida, Georgia, and Alabama remain dry, Louisiana had heavy rains last week, and scattered showers were predicted for this week. The weather appears ideal for rust development.
Late last October rust appeared in Indiana. The spores that gave rise to these infections most likely came from Louisiana, carried by southerly winds over a 2 to 3 day period in late September.
The rust that is now in Louisiana does not pose an immediate threat to Indiana because our soybeans are just being planted. If the rust persists and continues to develop in the Mississippi Delta, a storm system later in the summer could carry spores up into Indiana while our crop is vulnerable. If a spore dispersal event were to occur anytime from June through August, the disease could have time to develop here enough to cause damage.
In addition to presence of rust in the Mississippi Delta, there are some other variables that will determine whether rust becomes a problem in Indiana this year. Even though winds may carry spores from Louisiana into Indiana, some will die during transit. If few spores survive the journey, then disease may not develop. Even if there is some infection of Indiana soybeans during the summer, if local weather is not favorable for infection, rust will spread slowly. It is impossible to say at this time what our risk is, but the fact that rust is now in Louisiana suggests that our risk is greater than it was during the past two growing seasons.
We will continue to monitor rust development in the Delta region and watch for weather patterns that could bring spores north. Many soybean sentinel plots have already been planted in Indiana and will be scouted regularly for rust. We will also monitor several kudzu sites in the state.
Additional information about the status of rust can be found on the USDA PIPE Website, available at <http://www.sbrusa.net>. Purdue also maintains a toll-free soybean rust hotline, which I update regularly during the growing season. The phone number is 866-458-RUST (7878).
Root Development in Young Corn - (Bob Nielsen)
Successful emergence (fast & uniform) does not guarantee successful stand establishment in corn. The next crucial phase is the establishment of a vigorous nodal root system. Success is largely dependent on the initial development of nodal roots from roughly V2 (2 leaves with visible collars) to V6.
Corn is a grass and has a fibrous type root system, as compared to soybeans or alfalfa that have tap root systems. Stunting or restriction of the nodal root system during their initial development (e.g., from dry soil, wet soil, cold soil, insect damage, herbicide damage, sidewall compaction, tillage compaction) can easily stunt the entire plant’s development. In fact, when you are attempting to diagnose the cause of stunted corn early in the season, the first place to begin searching for the culprit is below ground.
To better understand rooting development and problems associated with root restrictions, it is important to recognize that root development in corn occurs in two phases. The first phase is the development of the seminal or seed root system. The second phase is the development of the nodal or crown root system.
The Seminal (Seed) Root System
Seminal (seed) roots originate from the scutellar node located within the seed embryo and are composed of the radicle and lateral seminal roots. The radicle emerges first and elongates towards the tip end of a kernel. The lateral seminal roots emerge later from behind the coleoptile and elongate towards the dent end of a kernel.
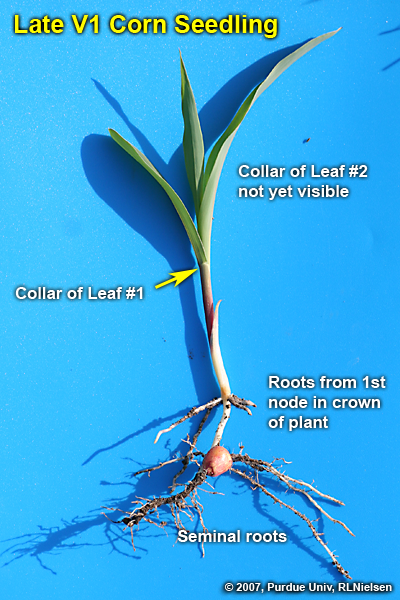
The seminal root system anchors the young seedling and absorbs small amounts of water and nutrients for the first two to three weeks. The rate of growth for these seminal roots slows down dramatically shortly after the coleoptile emerges from the soil surface. Conventional wisdom (or agronomic legend) suggests that though the seminal root system is important during these early stages of development, their contribution toward the season-long support of the plant is minimal if not nil.
A young corn seedling depends primarily on the energy reserves of the kernel until the nodal roots develop. These energy reserves are translocated from the kernel through the connecting mesocotyl “pipeline” to the young stalk and leaf tissues. Therefore, healthy kernel, seed roots, and mesocotyl are vital until the nodal roots are well established.
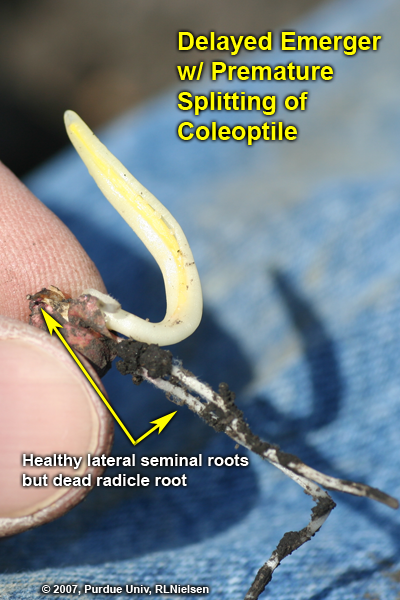
If damage occurs to seminal roots or the mesocotyl before the later-developing permanent roots become established, stunting or death of the plant will occur. Examples of such damage include salt injury from excessive rates of starter fertilizer, seedling diseases, herbicide injury and insect feeding damage.
Nodal (Or Permanent) Root System
Shortly after seedling emergence, the first set or whorl of the so-called permanent roots begins to elongate from the first of the stalk nodes located near the crown of the young seedling and are distinctly visible by at least leaf stage V2. Individual sets of nodal roots develop sequentially over time from each belowground stalk node plus one or more aboveground nodes. By leaf stage V6, five sets of nodal roots are typically well established and have completely taken over the sustenance of the plant.

Elongation of the stalk tissue begins between leaf stages V4 and V5. Elongation of the internode above the fifth node usually elevates the sixth node above ground. Subsequent elongation of higher-numbered stalk internodes will result in higher and higher placement of the remaining stalk nodes. Sets of nodal roots that form at above ground stalk nodes are commonly referred to as “brace” roots, but function identically to those nodal roots that form below ground. If surface soil conditions are favorable (moist and not excessively hot), brace roots will successfully penetrate the soil, proliferate, and effectively scavenge the upper soil layers for water and nutrients.
Corny Trivia: Root hairs are lateral extensions of root epidermal cells, grow to a length of several millimeters, and number about 200 per sq. millimeter (Gardner et al., 1985). Their typical life span is only about 2 days at moderate temperatures and less so at higher temperatures (Gardner et. al., 1985). Root hairs are visible even on the radicle root of a young seedling. Collectively, the surface area represented by root hairs is very large and can account for a large share of nutrient and moisture uptake by the plant.

A split stalk of an older plant will reveal a “woody” triangle of stalk tissue at the bottom of the corn stalk. This triangle is typically comprised of four stalk nodes, one on top of the other, whose associated internodes do not elongate. The first internode to elongate is the one above the fourth node, which elongates about 1/4 to 1/2 inch, above which is found the fifth node (usually still below or just at the soil surface). Consequently, five sets or whorls of nodal roots will usually be detectable below ground (one set for each of the below ground stalk nodes).
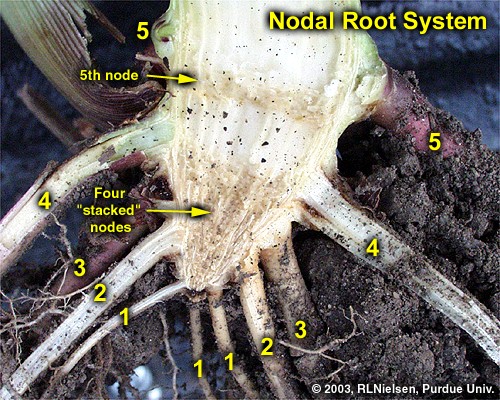
Effects of Root Damage in Young Corn Plants
Even though the seminal root system contributes little to the season-long maintenance of the corn plant, early damage to the radicle or lateral seminal roots can stunt initial seedling development and delay emergence. Such damage will not necessarily cause immediate death of the seedling as long as the kernel itself and mesocotyl remain healthy, but may result in the seedling leafing out underground. As more and more nodal roots become established over time, damage to the seminal root system will have less and less impact on seedling survival.
Examples of seminal root damage include imbibitional chilling injury, post-germination injury from lethal or sub-lethal cold temperatures, and “salt” injury from excessive rates of starter fertilizer placed too close to the kernel. Symptoms of such root damage include retarded root elongation, brown tissue discoloration, prolific root branching, and outright death of root tissue. If the radicle root is damaged severely during its emergence from the kernel, the entire radicle root may die. Once the radicle has elongated a half-inch or so, damage to the root tip will not necessarily kill the entire root, but rather axillary root meristems may initiate extensive root branching in response to damage to the apical meristem.
Corn seedlings transition from dependence on kernel reserves to dependence on nutritional support by the nodal roots around the V3 leaf stage. Damage or stress to the first few sets of developing nodal roots during the time period V1 to V5 can severely stunt or delay a corn plant’s development. Damage to the first few sets of nodal roots forces the young seedling to continue its dependence on kernel reserves longer than is optimum. If the kernel reserves are nearly exhausted, continued seedling development is easily stunted and seedling death is not uncommon. Typical stresses that can stunt initial nodal development include fertilizer salt injury, seedling diseases, herbicide injury, insect feeding damage, excessively wet or dry soils, soil compaction (tillage or planter).

Corny Trivia: The primary meristem of a root is located near the root tip. Elongation of cells behind the meristem leads to elongation of the root.
A somewhat uncommon, but dramatic, stunted root symptom is what is referred to as the “floppy corn” or “rootless corn” phenomenon. This problem occurs as a result of the detrimental effects of excessively dry surface soil near the time of initial nodal root elongation in young (V2 to V4) corn plants. Young nodal roots that emerge from the crown area of the plant will die if their root tips (and associated meristematic areas) desiccate prior to successful root establishment in moist soil. The crown of a young corn plant is typically located only 3/4 inch or so below the soil surface and so is particularly vulnerable to dry upper soil conditions.

Related References
Cannon, William Austin. 1949. A Tentative Classification of Root Systems. Ecology 30[4], 542-548.
Gardner, Franklin P., R. Brent Pearce, and Roger L. Mitchell. 1985. Physiology of Crop Plants. Iowa State Univ. Press, Ames, IA.
Nielsen, RL (Bob). 2004. Over-Extended Mesocotyls and Floppy Corn Syndrome. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/articles.04/FloppyCorn-0624.html>. (URL verified 5/16/07).
Nielsen, RL (Bob). 2007a. Germination Events in Corn. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/GerminationEvents.html>. (URL verified 5/16/07).
Nielsen, RL (Bob). 2007b. The Emergence Process in Corn. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/Emergence.html>. (URL verified 5/16/07).
Nielsen, RL (Bob). 2007c. Visible Indicators of Germination in Corn. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/GerminationGallery.htm>. (URL verified 5/16/07
For other Corny News Network articles, browse through the CNN Archives at <http://www.kingcorn.org/news/archive.html>.
For other information about corn, take a look at the Corn Growers’ Guidebook at <http://www.kingcorn.org>.
Crop Diagnostic Training and Research Center – (Corey Gerber)
The Purdue University Crop Diagnostic Training and Research Center is known across the Midwest for its unique “hands-on” approach for teaching the art and science of accurately diagnosing agricultural crop problems. The Center, established in 1985, is designed to provide informative topics in a “real world” environment, where agriculturists can sharpen their crop problem trouble-shooting skills and evaluate new and alternative management strategies. The long-term goal of the Center is to provide quality, state-of-the-art training in all aspects of crop production and management.
At this Center, many small plot demonstrations illustrate insect, nematode, weed, disease, soil fertility, and cultural problems associated with corn, soybean, forage, and small grain production. Extension, research, and teaching staff from Purdue University’s departments of Agricultural and Biological Engineering, Agronomy, Botany and Plant Pathology, and Entomology, as well as other leaders in the agricultural community across the U.S., conduct each workshop.
Please visit the Calendar section of this Web site to view workshop dates, agendas, speakers, and the CCH’s & CEU’s available for each workshop.
The Purdue University (West Lafayette) Crop Diagnostic Training Center is located on the grounds of the Purdue University Agronomy Center for Research and Education.
For additional information on specific training days, costs, housing, and registration information visit the website: <http://www.agry.purdue.edu/dtc/index.html> or contact:
Corey K. Gerber, Ph.D., Director
Purdue Crop Diagnostic Training and Research Center
Purdue University, Agronomy Department
915 W. State St.
West Lafayette, IN 47907-2054
(765) 496-3755