Pest & Crop Newsletter, Entomology Extension, Purdue University
Early Planting and Rootworm Insecticide Efficacy – (John Obermeyer, Christian Krupke, and Larry Bledsoe)
- Rootworm egg hatch may have begun in southern Indiana, still a week or two away for the rest of the state.
- Soil insecticides applied with early-planted corn are subject to dgradation and/or off-target movement by the time they are needed.
- Larger root systems of early-planted corn may icrease larval survival.
- Rootworm larval sampling is encouraged for these high-risk fields in mid-June.
This year’s corn planting is just behind last year’s record pace according to Indiana Agricultural Statistics. By the third week of April, over 35% of the state’s corn had been planted, much of that in high rootworm risk areas (Pest&Crop #27, 2004 www.entm.purdue.edu/Entomology/ext/targets/p&c/index2004.htm). With rootworm egg hatch yet to occur in a week or two in central and northern Indiana, how effective will the granular, liquid, and seed-applied insecticides be after “sitting” for so long in and on the soil?
Remember that soil insecticides do not kill rootworm eggs, only hatched larvae. Rootworm egg hatch is mainly dictated by soil temperature, and normal egg hatch in high-risk rootworm areas of the state occurs in late May or early June. Because of our widely fluctuating air temperatures this spring, we would not expect an early hatch this season. As a result, about 1/3 of Indiana’s corn is being protected by an insecticide that was applied 5 to 6 weeks before it is needed.
All soil/seed insecticides begin to degrade once applied in or on the soil surface. The two main factors that break down rootworm insecticides are moisture and sunlight. High moisture levels move the insecticides deeper into the soil profile, sometimes too far. However, those fields that received “toad floaters” may have either physically moved the insecticide off-target or caused deeper movement into the soil. The sun’s UV rays will degrade insecticide exposed on the surface of the soil. This is particularly important with T-band applications that can leave a considerable percent of product exposed if there wasn’t any incorporation (e.g., drag chains). Finally, heavy crop residues will hinder a portion of the product from even making it to the soil.
Another consideration is that early-planted corn typically has a larger root system by early June. Though this is a good thing for the plant’s moisture and nutrient uptake, it may also support higher rootworm survival. Hatching rootworm larvae have a limited time and ability to crawl through the soil to find a corn root before starving. As we saw last year, saturated soils can hinder this movement and even cause drowning. However, larger root systems will increase the likelihood of rootworms finding their food source. Because soil insecticides tend to move downward rather than laterally (the opposite of corn root growth patterns) in the soil profile, root systems may grow beyond the protected band of soil insecticides, leaving them vulnerable to feeding.
By mid-June, pest managers should consider inspecting these early-planted fields for rootworm damage, especially those subjected to environmental extremes in a high rootworm risk area. In future issues of the Pest&Crop, we will have updates of rootworm hatch, larval sampling procedures, and reports of damage. Happy scouting!
![]()
Planning For the 2005 Soybean Aphid Population – (David Voegtlin, Center for Ecological Entomology, Illinois Natural History Survey; and Robert O’Neil, Entomology, Purdue University)
- 2004 fall sampling of soybean aphid migration to buckthorn suggests higher numbers than last year.
- Many factors in the spring, some yet to be understood, determine soybean aphid densities for the growing season.
- Soybean aphid has not yet been found on soybean in the Midwest.
- Successful aphid migration to soybean this spring does not suggest economic populations are looming this summer.
- Properly scout and use treatment thresholds to manage this potential pest in 2005.
With soybean planting well underway for 2005 there is a lot of concern about potential pest problems, both rust and aphids this year. Following a year in which the soybean aphid was widely distributed in the Midwest but rarely common enough to generate concern, the question is what about 2005?
There are a number of factors that come into play that determine aphid numbers. The soybean aphid survives the winter as eggs on common buckthorn (scientific name: Rhamnus cathartica), an exotic, weedy, shrub common in much of the Midwest north of I-80. In the spring these eggs hatch and several generations on buckthorn produce winged aphids that migrate in search of soybeans. For the past four years a suction trap network has been operating in Illinois. While we have been unable to monitor the spring movement of aphids from buckthorn to soybean, we have been able to monitor the fall flight of aphids from soybean to buckthorn.
In 2001 and 2003 we had low fall flights and the next growing year the soybean aphid was not a problem. In 2002 we had a large fall flight and we experienced a major and widespread outbreak of the aphid in the 2003 production season. The fall flight in 2004 was the highest yet, begging the question: “Does the large fall flight of 2004 mean that 2005 will be a repeat of 2003?”?
This question is difficult to answer because many factors can influence the size of the following year’s aphid population in soybeans. To start with, large numbers of multi-colored Asian lady beetles can be found on shrubs and trees in the latter part of September and into October. An abundance of these predators on buckthorn can effectively prevent the deposition of over-wintering eggs, and the subsequent production of spring migrants that fly into soybeans. Those eggs that are successfully deposited must survive the winter and after successfully hatching, they need to survive the spring weather. Heavy rains and sub-freezing temperature in early spring can eliminate young colonies on buckthorn. In the spring, lady beetles (and other predators) become active and will feed on soybean aphid colonies on buckthorn. Timing of planting can also impact the success of the spring migration. Outdoor cage tests and field observations in central Illinois have shown that many winged aphids leave buckthorn in late April and early May. This migration often precedes the planting and emergence of much of the soybean crop. (This gap between the migration of winged aphids from buckthorn and the availability of soybeans decreases in the northern growing regions and increases in the south.).
Finally, a spring migrant successfully finding soybeans does not necessarily guarantee the development of a colony. Studies by various university researchers have shown that a number of natural enemies, both predators and parasites, are present in soybean fields. When these natural enemies are eliminated or excluded in field experiments, soybean aphid colonization is more successful and population growth is rapid. So while the aphid can outbreak, the factors that lead to an outbreak are many, and all along the way lay pitfalls that prevent or ameliorate aphid outbreaks in any given year.
Spring of 2005 field observations: There have been a number of reports from throughout the Midwest of aphids on Rhamnus. Dr. Ho Jung Yoo of Purdue has been monitoring colonies in Indiana. Dr. Ron Hammond at The Ohio State University documented egg hatch around the last week of March. Dr. Christina DiFonzo from Michigan State University noted egg hatch in early April. In Illinois, we have found colonies in the Quad cities and in the Rockford area as well as in Champaign.
But how do we interpret these findings?? It seems inevitable that there will be more aphids than in 2004, but will populations reach the pest levels seen in 2003?? The best we can say now is maybe. We now have an economic threshold (250 aphids/plant), and farmers are now well aware of the damage potential of the soybean aphid. Unlike in 2003 when farmers did not realize the impact the aphids were having on their plants (thus allowing their populations to grow), it is unlikely that growers will tolerate a similar build up of aphids this year. The local control of aphids may, therefore, limit the production of large numbers of winged aphids that would fuel a widespread outbreak. However, on the flip-side, chemical control measures against populations reaching the threshold level can wipe out the local predators so that any winged aphid arriving in one of these sprayed fields will experience less resistance to successful colonization. Likewise the aphids surviving an application would have few natural enemies left to prevent aphid population build-up and the field may experience a “rebound” effect leading to faster aphid population growth and pest damage.
So what can farmers do?? Farmers should know that there are lots of people looking for the aphid and they are networked. News of infestations travel fast and farmers should have good lead-time, particularly if they keep in touch with their local Extension Service, fellow farmers and state Land Grant Universities.
Second, farmers should be scouting for aphids. A weekly visit to their fields to examine even a few plants will help them keep abreast of aphid population growth in their crop. Third, farmers should use the threshold of 250 aphids per plant. To use the threshold properly, it’s important to know if the aphid population is increasing and that’s why farmers need to visit fields regularly to look for aphids. And fourth, farmers should not panic! Just because they have some aphids in their field does not mean they will reach the threshold of 250 aphids per plant. The natural enemies in their field are providing free pest control in most fields and in most years. Spraying fields before the threshold only helps to disrupt the control these natural enemies provide and does not guarantee economic control. The temptation to “tank-mix” a little insecticide with an herbicide (or fungicide) application “to get those aphids before they get me” is to be avoided! Looking for aphids, checking the threshold, and then deciding if you need to do something, is the key for this year’s aphid pest management.
![]()
Slug Damage and Rescue Controls – (John Obermeyer, Christian Krupke, and Larry Bledsoe)
- Slugs are favored by a wet spring with heavy crop residue on the soil surface.
- Crop damage and stand losses are most severe when slugs enter open seed slots.
- Finding slugs during the day can be difficult.
- Control options are limited.

Slug damaged hypocotyl.
We have received one call on slugs damaging emerging soybean this past week. With slugs, one call indicates that other fields of soybean and corn are being damaged but haven’t been identified. Soybean damage is usually manifested as gouges on the hypocotyl and cotyledons that may cause plant death. On corn, slugs feed on the surface tissue of leaves resulting in narrow, irregular, linear tracks or scars of various lengths. Given good growing conditions, plants usually outgrow slug damage once the crop is up. Most stand losses by slugs occur in areas of the field where seed slots were not properly closed. In this situation, slugs can be found feeding on the seedlings within the slot, day or night. Obviously, once the growing point of corn or soybeans is injured, plant recovery is unlikely.
Confirming that slugs are causing the damage can be difficult during daylight, because they are nocturnal feeders except when feeding in dark seed slots. Their damage can be confused with other insect damage (e.g., flea beetle, small cutworm, etc.). Glistening slime trails found on the damaged crop or nearby soil surface are good indicators of slug presence. To find the slugs, pull back crop residues from the soil surface near the row and look for slugs balled up in moist areas. Digging in and around the soil of injured plants, especially in the seed slots, may reveal slugs that have taken cover during the day. To make certain that you indeed have a slug, place the gooey ball on your hand and watch to see if it uncurls and slithers away. It’s fun!
Chemical control of slugs is difficult, if not impossible. Metaldehyde pelleted baits, Deadline M-Ps and Trail’s End LG, are labeled and available for use. The cost for product is about $10-15 per acre. Spreading the pellets evenly over the field or damaged areas is key to control; using a commercial mechanical dispenser is one possibility. Field trials at Ohio State University have shown promising results when the pellets are evenly distributed. With the significant cost and difficulty of application, consider these baits only as a last resort to protect crop stands in high slug populated areas.
Where replanting is necessary from slug damage, one should strongly consider tilling (disc and/or field cultivator) the area first. This should help dry the area and break-up and bury crop residue. Doing so will discourage further slug activity. Granular and liquid insecticides are ineffective against slugs, as slugs secrete slime to protect themselves from these products.
![]()
Click for Table.
Black Cutworm Adult Pheromone Trap Report.
Click for Table.
Black Light Catch Report.
![]()
Weed and Volunteer Corn Management in Replanted Corn Fields - (Bill Johnson and Glenn Nice)
We have received several questions about weed and corn management in replanted corn fields. Here are a few important points to consider.
The best weed management strategy in these fields will be to scout and spray postemergence herbicides. It is difficult to know how much residual herbicide is left in fields planted in early to mid april and soil applied herbicides have caps on the maximum amount of product that can be applied in a season. So, with the plethora of postemergence herbicides available for corn, we should be pretty successful at post weed control if herbicides are applied in a timely manner. Corn is now growing rapidly and corn growth stages must be monitored to avoid applications past the stages listed on the label. For a summary of maximum heights and growth stages with specific herbicides, see Table 7 in the Weed Control Guide for Ohio and Indiana www.btny.purdue.edu/Pubs/WS/WS-16/CornRainfast05.pdf.
Killing the 1st corn stand in a replant situation. If 1st stand was not Roundup Ready one can use low rates of glyphosate (11 oz/A of Roundup Weathermax or 16 oz/A of other 3 lb ae/gallon products). This treatment will be most effective on corn that has 2 or more exposed leaves to maximize spray coverage. Obviously this treatment would need to be applied before the new corn emerges. If the 1st stand was Roundup Ready corn the situation is much more difficult. Post grass herbicides for soybean will generally have too much residual activity in soil to allow their use and it is unlikely that any of the manufacturers will stand behind their use. Consider using high rates of Gramoxone with some atrazine (if the field has been replanted and the new stand has not emerged) or replanting with Liberty Link corn and applying Liberty when the old stand has 2 or 3 fully developed leaves, or applying Balance to the emerged old stand, but before the new corn emerges. A Gramoxone treatment may be more effective if the corn has at least 3 or 4 exposed leaves. Treatments made too early might just burn off the top of the plant and not kill the growing point. Finally, tillage can be utilized to tear up an old stand, but may cause compaction, loss of soil moisture, and it may disrupt or dilute activity of previously applied soil-applied herbicides.
Dry conditions in Northern Indiana. Dry weather in parts of northern Indiana have resulted in a number of instances where weeds are breaking through soil applied herbicides. Rotary hoeing can still be used on some later planted corn if the weeds are just starting to poke through the ground (white stage). In early planted fields with larger weeds (1 inch tall or more), post herbicides will be the best choice at this time![]()
Sugar Maple in No-Till Fields – (Glenn Nice and Bill Johnson)
- Identification
- Control options

Figure 1. Mature sugar maple tree.
Photo credit: S. Weeks "Trees of Indiana" (CD-FNR-1).
No-till fields sometimes have problems with perennial weeds like common pokeweed, Johnsongrass, and on occasion tree species. One species that we have noticed in several no-till fields over the past couple of years is sugar maple (Acer saccharum Marsh [Figure 1]) seedlings.
Identification
The sugar maple is a native of Eastern US that has been planted as an ornamental in many yards and along roads. Its fame comes from using its sap for maple syrup, but it has also been used for its wood. For Canadians it is significant in the fact that it is a major component of the Canadian flag. It grows to be a large tree of 100 to 150 ft tall with a trunk diameter up to 2.5 to 3.5 ft. It has a typical maple leaf 3 to 6 inches long with 3 to 7 lobes (Figure 2). Leaves are generally dark green on the top and light green on the underside. Sugar maples flower in April to May. Their flowers are greenish yellow and droop on hairy pedicels. Even at a small size the seedling will show the typical leaf shape.

Figure 2. Sugar maple leaves showing lobes.
Photo credit: S. Weeks "Trees of Indiana" (CD-FNR-1).
Although these are beautiful trees they are generally not welcome in a soybean or corn field. If allowed to persist they become more troublesome to control in a row crop field. The papery wings of the seed can carry seed over 300 feet from the parent tree, easily across a road or into your field. Seedlings can germinate and do well in either shade or full light, emerging in April and May. When seedlings emerge, much of the season’s growth occurs with 24 days of emergence.
Control options
Information for the control of sugar maple tends to revolve the control of the small or large trees in a forestry setting. When doing a search of labeled products in corn or soybean, generally only glyphosate products appeared to be labeled to control sugar maple. The recommendations on most labels refer to a spot treatment when at least 50 percent of the leaves are present. These applications are applied using a 0.75 to 1.5 (rates vary depending on label) percent solution and a hand held back pack sprayer. Some dicamba (Banvel) labels address cut surface and stump applications for the control of maples and Clarity’s label has maple as a control species. However, information to the specific control of sugar maple seedlings in corn or soybean was not fully addressed. Considering the above mentioned products do have activity on larger mature trees, we suspect that both glyphosate (Roundup) and dicamba (Banvel, Clarity, Distinct products) are effective on sugar maple seedlings, but one thing is for sure, the topic is definitely open for some investigation. Our recommendations would be to use the highest allowable rates of these products in the burndown or postemergence in the crop. If they are established in a long-term no-till field, consider aggressive tillage of the specific area where the trees are growing or fall mowing and cut stump treatments if tillage is not desirable.
Reference:
Britton, N.L., and A. Brown. 1913. Illustrated flora of the northern states and Canada. Vol. 2: 496.
Sally S. Weeks and George R. Parker. Trees of Indiana, Their Identification and Uses. Purdue University Agricultural Communications interactive cd. CD-FNR-1
![]()
Anthesis: Critical Stage for Soft Red Winter Wheat — (Shawn Conley and Gregory Shaner)
- Weather at anthesis can have a big effect on yield and quality.
As the winter wheat crop develops in Indiana, it is critical that growers continue to monitor the crop. Based on the location within the state, the wheat crop currently ranges from anthesis, or flowering, (Feekes 10.51) in southern Indiana to boot swollen (Feekes 10) in northern Indiana. Anthesis is signaled by extrusion of anthers from each floret of the wheat head (Image 1.). Anthesis starts in the center of the head and progress toward each end of the wheat head. Completion of anthesis generally takes 3 to 5 days depending upon the variety and weather conditions. At this crop growth stage, wheat is very sensitive to environmental stress (hot or cold temperatures). At harvest, if a grower notices that either the center or the ends of the wheat head are barren, most likely an environmental stress occurred at anthesis.

Image 1. Soft red winter wheat at anthesis (flowering) (Feekes: 10.51, Zadoks: 60).
Anthesis is also a critical period for infection by Gibberella zeae (Fusarium graminearum), the fungus that causes Fusarium head blight, or scab, of wheat. In the Corn Belt, this fungus survives the winter in corn residue, and then produces spores during late spring. These spores can infect wheat and cause head blight. Extruded wheat anthers seem to be the favored site of infection. From the anthers, the fungus invades the head and developing kernels, which results in premature bleaching of the head and scabby grain. Production of spores on corn residue and infection of wheat require prolonged wet, warm weather. Our weather this spring has likely not been favorable for head blight. A weather-based risk model www.wheatscab.psu.edu/riskTool.html indicates low risk for wheat that began flowering any time from May 10 through the present, for fields that are not planted into corn residue. For fields planted into corn residue, the predicted risk is greater in southwest Indiana for some flowering dates.
Anthesis is a critical growth stage for timing of foliar fungicides. None of the available products is labeled for application past Feekes 10.5. It is therefore critical to monitor the wheat crop between full emergence of the flag leaf and anthesis, to determine whether a foliar fungicide is necessary, and if so, to apply it at the correct time. In general, disease pressure seems to be light this year, not only for head blight but for foliar diseases, too.
![]()
Tips for Staging Damaged Corn – (Bob Nielsen)

V3 corn seedlling with lower leaves damaged by frost/freeze.

Closer view of leaf collars of damaged V3 seedling.

Closer view of leaf collars of damaged V3 seedling.

V3 corn seedling with #3 leaf marked for future leaf staging assistance.
The current debate amongst the regulars down at the SunShine Café centers on the burning question of how to identify the age of a corn plant that has recovered from rather severe leaf damage. Some may wonder why this topic is worthy of vigorous debate. The reason lies with the fact that a number of post-emergence corn herbicides can only be applied to corn younger than a label-specified leaf stage (Table 7 in 2005 Weed Control Guide for Ohio and Indiana www.btny.purdue.edu/Pubs/WS.WS-16/CornRainfast05.pdf. Growers and applicators obviously need to be able to accurately stage corn plants to determine whether they are still within the herbicide label restrictions.
The issue for debate stems from the fact that many early-April planted corn fields suffered moderate to severe leaf injury from several frost/freeze events in late April and early May. The lowermost one to three leaves were often killed outright, but the plants usually recovered from the damage once sunshine and warm temperatures returned. Unfortunately, the dead leaf tissue does not recover and will eventually slough off as the plants continue to grow.
The hotly debated question is whether the leaf stage of a recovered plant begins anew with the healthy leaves or whether the dead leaves (which may no longer be visible) should be counted. In other words, should a 5-leaf plant that has lost two lower leaves to frost injury now be considered only a 3-leaf plant?
The answer is: A 5-leaf plant is a 5-leaf plant no matter how many lower leaves are damaged, dead, or otherwise missing.
As long as the dead lower leaves remain attached to the plants, leaf staging is reasonably simple. Count the dead leaves and any additional ones with visible leaf collars (Nielsen, 2004a). The challenge occurs when the dead lower leaves slough off and decompose or blow away. Now how do you count leaves if you are not sure whether the lowermost remaining leaf is #2 or #3? This problem is easily solved when staging V7 or older corn because one can then split stalks to identify the 5th node and its respective attached leaf (Nielsen, 2004a). Because stalk development is essential nil for corn plants V5 or younger, this strategy for staging corn does not work for younger plants.
Here are two alternatives for staging damaged young corn:
- Walk damaged and recovering fields soon while dead lower leaves of damaged plants are still attached. Mark ten consecutive plants with plot flags or garden stakes. Identify & record the leaf number of the lowermost healthy leaf (one likely to remain attached for some time) of each plant. Mark each such leaf by simply ripping off a third or half of the leaf. To simplify future leaf staging, mark the same leaf number on each of the 10 consecutive plants. When you come back in a week or later to stage the plants again, find the marked leaf on each plant and continue counting the number of leaves with visible leaf collars.
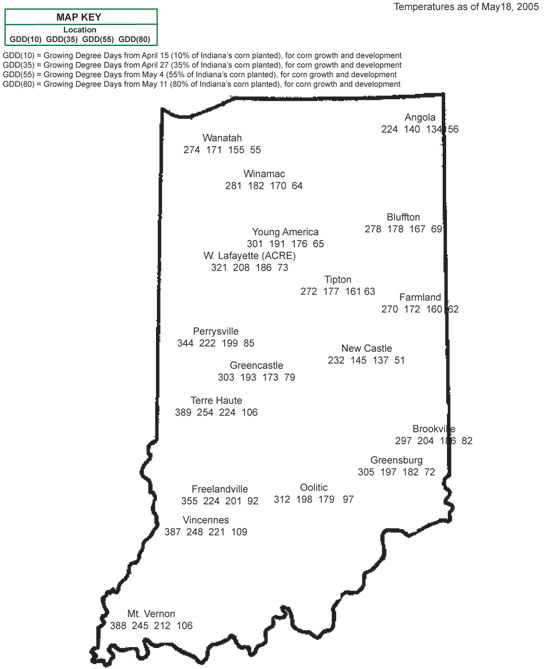
- Predict leaf stage development based on thermal time (growing degree days or GDDs) from planting or emergence. The relationship between corn development and temperature is reasonably strong. Emergence requires about 125 GDDs from planting and leaf collar emergence (up to about leaf #10) requires about 85 GDDs per leaf. See my article from last year for more details on how to predict leaf collar development based on thermal time (Nielsen, 2004c). Current year GDD data for various areas of Indiana are available online at the Indiana Climate Page http://shadow.agry.purdue.edu/sc.obs-geog.html.
Related References
2005 Weed Control Guide for Ohio and Indiana (WS-16). 2005. Table 7: Rainfast Intervals, Spray Additives, and Maximum Crop Size for Postemergence Corn Herbicides. Purdue Univ. & Ohio State Univ. Coop. Ext. Services. Available online at www.btny.purdue.edu/Pubs/WS/WS-16/CornRainfast05.pdf [URL verified 5/17/05].
Nielsen, R.L. (Bob). 2004a. Determining Corn Leaf Stages. Corny News Network, Purdue Univ. Available online at www.kingcorn.org/news/articles.04/VStageMethods-0515.html [URL verified 5/12/05].
Nielsen, R.L. (Bob). 2004b. Heat Unit Concepts Related to Corn Development. Corny News Network, Purdue Univ. Online at www.kingcorn.org/news/articles.04/HeatUnits-0515.html. [URL verified 5/12/05].
Nielsen, R.L. (Bob). 2004c. Use Thermal Time to Predict Leaf Stage Development in Corn. Corny News Network, Purdue Univ. Online at www.kingcorn.org/news/articles.04/VStagePrediction-0515.html [URL verified 5/12/05].
For other Corny News Network articles, browse through the CNN Archives at www.kingcorn.org/news/archive.html.
For other information about corn, take a look at the Corn Growers’ Guidebook at www.kingcorn.org.
Bug Scout

"Who said we should scout this field before that little ol' cloud got here?"
![]()