Pest & Crop Newsletter, Entomology Extension, Purdue University
Grub Problems: Wait and See – (John Obermeyer, Christian Krupke, and Larry Bledsoe)
- Early corn planting and cool soils increase the likelihood of grub damage.
- Soil type is another important variable.
- No rescue treatments are available for economic populations.
- Insecticides labeled for grub control or protection are listed below.
According to Indiana Agricultural Statistics’ most recent crop report, corn planting is almost identical to last year’s record pace. Grub complaints typically increase in frequency during an early planting season. We consider planting before the third week in April to be early. However, last year we heard of very few grub problems. Obviously there’s more to it than just planting date. Factors such as the field’s grub populations and soil type coupled with spring growing conditions all play a part.
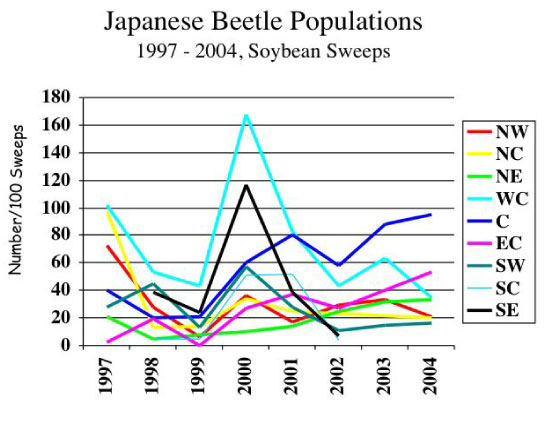
Japanese beetle is the predominant grub species found in field crops in Indiana. Eggs laid last summer and fall in the soil hatch into grubs that feed on living and decaying plant matter. Grubs overwinter as partially developed larvae about 4 to 6 inches deep in the soil. Little is known or understood about their ability to withstand extremes in soil temperature, moisture, and freezing/thawing action through the winter months. We believe there is a correlation between colder than normal fall and winter soil temperatures and fewer Japanese beetle adults the following summer. Refer to the graph below showing Japanese beetle numbers from soybean sweeps taken in the years 1997 to 2004. Clearly, statewide beetle numbers have been low during the last several years, though it is important to note that some local populations can be very large.
Seed that is already planted will likely be subjected to cooler soils and extended germination/emergence. If corn is slow to emerge and grubs are found nearby, it is often assumed that they are feeding on the seed/seedling. However, the reason for slow germination and emergence is usually cool soil temperatures, and not grub activity. Grubs may or may not be damaging the crop because they too are less active in cool soils. Once soils warm up … you can bet grubs will feed on roots but they are also feeding heavily on organic matter in the soil too. The length of the feeding period and grub population will largely dictate whether economic damage will occur. In other words, the longer a grub is near the seedling, the greater the likelihood of damage. As indicated above, the chances of this interaction increase as soil temperatures decrease.
| Products Labeled for Grubs at Planting* | ||
| Product | Label Claims | Additional Label Notes |
| Aztec 2.1 & 4.67G | control | |
| Capture 2 EC | control | |
| Cruiser | protection | Early season protection |
| Force 3G | control | Use higher label rate in-furrow for heavy infestations |
| Fortress 2.5 & 5G | control | In-furrow application provides optimal control |
| Lorsban 4E | control | |
| Lorsban 15G | control | Control at 1.5X rootworm rate for severe infestations. |
| Poncho | protection | Aid in the protection of seeds and seedlings against injury |
| Regent | control | |
| * Products labeled for grubs often do not perform satisfactorily under heavy infestations. If grubs are causing economic damage in fields where products labeled for "control" are used, producers should be contacting their dealer and/or sales representative for a performance evaluation. Producers should be cautious using products labeled "protection" where higher grub pressure is expected. Be sure to read the label for use and application information. | ||
Japanese beetle grubs feed on both living and dead material when they crawl to the upper soil profile in the spring. Soils low in organic matter (O.M) and crop residues will encourage grubs to move more horizontally in the soil profile until suitable food sources are found. Corn or soybean roots that they encounter will certainly be fed upon. Should you scout a field suspected to have grub damage, be certain to inspect the soil between rows for grub presence. There you will likely find as many, if not more, grubs in the soils that have significant organic material. However, grubs in sandy or timber soils (i.e., low O.M.) will concentrate in root zones because alternate food sources are scarce.
Since rescue treatments are not available, the most effective way to control grubs is to apply protection at planting (see table below). If an economically damaging grub population is observed in a field that has already been planted and the stand is threatened, a soil insecticide/insecticide treated seed could be used as part of a replant operation. Replanting, however, is not recommended unless a critical level of plants is being significantly damaged or destroyed by grubs. Remember that a number of factors can cause stand reductions. Be certain that the grubs are the primary cause of damage, and are actively feeding on the roots before making a replant decision.
![]()
What Should We Expect from Nematodes This Spring? – (Jamal Faghihi and Virginia Ferris)

Needle nematode damage to corn.
A lot of growers have already planted their corn. In the past three years we had a cool and wet spring, which is the ideal condition for needle nematode to cause damage in corn. However, this has been a different season so far. A dry and warm spring usually makes life difficult for needle nematode. So, if these conditions persist we don’t anticipate encountering much problem from needle nematode on corn. However, if conditions happen to change to cold and wet in the next couple of weeks, while corn is germinating and beginning to grow, we might still have problem with needle nematode limiting growth of young corn seedlings. If you encounter patches of stunted young corn seedlings in sandy soil, needle nematode might be the problem. In this case, you need to send an entire root system with adjacent soil to the Nematology Laboratory at Purdue University for analysis. Samples must be kept cool and prevented from drying.
This spring, conditions have been ideal for soybean cyst nematode (SCN) early activity. The nematodes do not require the presence of soybeans to come out of the cyst and move in the soil. As soon as susceptible soybeans have germinated they are prepared to invade the young soybean seedlings. If this weather condition persists, we anticipate heavy damage to susceptible soybeans from soybean cyst nematode, especially in sandier soils. If you have not sampled for SCN in the past or used resistant soybean varieties for several years, you need to sample for this nematode. As in previous years, Indiana Soybean Board is paying the processing fees for Indiana growers, up to 10 samples/grower/year. You can sample for SCN anytime of the year regardless of the type of current or previous crops. Soil samples taken from a depth of 4-6 inches can be sent to our laboratory for analysis.
If you have any questions about these or any other kinds of nematode, you can call 765-494-5901 or send an email to jamal@purdue.edu. Soil samples for nematode analysis can be sent to: Nematology Laboratory, Purdue University, Department of Entomology, Smith Hall, 901 W. State Street, West Lafayette, IN 47907-2089. Go to our website
www.entm.purdue.edu/Entomology/ext/nematology.html to download the Nematode Sample Form and the instructions on How to Collect Samples for Nematode Diagnosis.
![]()
Click for Table.
Black Cutworm Adult Pheromone Trap Report.
Click for Table.
Black Light Trap Catch Report.
![]()
Poison Hemlock - The Toxic Parsnip - (Glenn Nice)
- Description
- Symptoms of poisoning
- Treatment
We often get questions about wild carrot (Daucus carota L.) only to find out that the question is actually about poison hemlock (Conium maculatum L.). Although these two plants may look similar, poison hemlock is toxic to cattle, horses, swine, sheep, goats, dogs, and people when ingested. The plant produces volatile alkaloids coniine (an alkaloid similar in effect to nicotine) and gamma-conicine. The easiest way to tell the two plants apart is that poison hemlock will have purple spots or blotches on its smooth (hairless) ridged stems. Wild carrot will usually have a covering of hairs.
Description:

Figure 1. First years growth of poison hemlock.

Figure 2. Second years growth, poison hemlock flowers in bloom.

Figure 3. Poison hemlock leaf.
Poison hemlock can often be found along roadsides, edges of cultivated fields, railroad tracks, stream banks, waste areas, and sometimes along the fence rows of pastures. Like wild carrot, poison hemlock is a biennial. This means that it lives its life over two years. In the first year, poison hemlock goes through vegetative growth (figure 1). In the second year, it will produce small white flowers arranged in umbrella-like cluster (figure 2), similar to wild carrot. It is in the second year, when it bolts and flowers, that it tends to catch the eye. The flower stalks can grow 3 to 8 feet tall. The leaves are finely divided having a triangular shape (figure 3). When comparing both poison hemlock and wild carrot leaves, wild carrot has a more rounded lobe in the leaf. Where as poison hemlock’s leaf reminds me of a sharp arrow head. For more information and pictures of poison hemlock’s description go to www.ppws.vt.edu/scott/weed_id/coima.htm and www.vet.purdue.edu/depts/addl/toxic/
plant28.htm.
Symptoms of Poisoning:
All parts of the plant can be toxic. Young leaves in the spring are the most toxic and the root the least toxic. The fruit is most dangerous in the fall. Lethal doses can be small, so it is important not to let animals graze or feed on poison hemlock. In the case of horses, 4 to 5 pounds of the leaves may be lethal. One to 2 pounds can be lethal for cattle and 4 to 8 oz for sheep. Young animals are more susceptible. Symptoms may appear within 1 hour of ingestion. This starts with a nervous stimulation and can progresses in 2 to 3 hours later into respiratory paralysis. In rare cases the animal may have convulsions. In many cases symptoms include, bloating, incoordination, intestinal irritation, dilation of pupils, rapid and weak pulse, loss of appetite, salivation, and blue coloration about the mouth. Ingestion of poison hemlock in days 55 to 75 of gestation may result in birth defects.
Treatment:
If an animal becomes poisoned by poison hemlock, a veterinarian may administer nerve and heart stimulants as soon as possible. Large doses of mineral oil and purgatives are also prescribed to empty the digestive tract. For more information about poison hemlock’s toxic characteristics please see <www.vet.purdue.edu/depts/addl/toxic/plant28.htm>.
Control of poison hemlock in grass pastures is more effective in the first year of its life cycle. In a grass pasture, 2,4-D, Banvel/Clarity (dicamba), or Crossbow (2,4-D and tryclopyr) provide control. Crossbow is slightly better than both 2,4-D and Banvel/Clarity. However, be aware that these herbicides will damage any legumes. Spot treatments of glyphosate products (Glyphomax Plus, Roundup WeatherMax, Touchdown, etc) will also control poison hemlock, but be aware that this will also damage any desired vegetation. Always read and follow labels when using herbicides.
We have also noted that poison hemlock is beginning to invade no-till corn and soybean fields. Herbicides that have activity on this weed and that can be used before planting soybeans are 2,4-D, dicamba, and glyphosate. Dicamba and glyphosate have shown slightly better efficacy than 2,4-D. The best overall control would likely be attained with a mixture of glyphosate and dicamba.
Considerations Using Dicamba in Soybean: Use 1/2 pt/A on coarse soils and 1 pt/A on medium or fine soils with at least 2% organic mater. Not all dicamba products have PRE-plant labels in soybean: consult the label before buying for this purpose. Clarity can be applied 14 days before planting if 8 fl oz/A or less is used and at least 1 inch of rainfall or over head irrigation occurs; however, if 16 fl oz/A is used there is a 28 days waiting period before planting soybean.
Considerations Using 2,4-D in Soybean: Use 1 to 2 pt/A of a LVE (Low Volatile Ester formulation). Before planting soybean, using 2,4-D at 1 pt/A requires a 7 day waiting period before planting and if more than 1 pt/A is used, a 30 day waiting period must be observed. These restrictions may be slightly different depending on the product, please read the specific products label before buying for this purpose.
Considerations Using Glyphosate: Use 0.75 lb ae/A.
Ae stands for acid equivalent. This is the glyphosate weight in its acid form. Most labels will give ae per gallon to calculate amount to be used. Glyphosate is a non-selective herbicide and can damage desirable vegetation if it comes in contact with desirable vegetation.
For more information on toxic plants of indiana to livestock and pest see www.vet.purdue.edu/depts/addl/toxic/cover1.htm.
![]()
A Risk Model for Fusarium Head Blight of Wheat is Up and Running - (Gregory Shaner)
A web-based risk model for Fusarium head blight of wheat is available again this year. It can be accessed at www.wheatscab.psu.edu/. Much like a weather forecast, the model predicts the probability of an event, in this case an epidemic of head blight. For purposes of this model an epidemic is defined as a head blight severity of at least 10% in the field. Severity is the product of the percentage of heads that are blighted, and the average percentage of spikelets on each head that are blighted. For example, severity would be 10% if 10% of the heads were completely blighted, if 20% of the heads were 50% blighted, if 100% of the heads were 10% blighted, and so forth.
Weather has a strong influence on development of this disease. The fungus that causes head blight, Fusarium graminearum, also causes Gibberella ear and stalk rot of corn, and is ubiquitous in corn residue. Although the fungus is abundant, if weather is unfavorable for spore production or infection during a critical period of wheat development, there will be little or no disease. Conversely, if weather is favorable, a major outbreak of head blight will occur, often over a large area. In each of the past 2 years, head blight has been common and widespread in Indiana and neighboring states.
At the homepage for this head blight risk model, a user can find background information about the disease as well as information about the model. To use the model, select the Fusarium Head Blight Risk Assessment Tool. From a calendar on the left side of the page, select the flowering date. This is the date at which plants reach Feekes growth stage (GS) 10.51 (See issue 2, March 25, of Pest&Crop for a description of wheat growth stages). It is important that flowering date be accurately assessed. Once wheat starts flowering, extruded anthers may be retained on heads for several days. Therefore, it is important to inspect a field regularly once heads are in the boot, to determine when flowering actually begins. Next, choose the model to run: winter wheat or spring wheat. When winter wheat is chosen, two other choices appear: corn residue present or not. In the development of this model, it turned out that presence or absence of corn residue in a field had a strong influence on the risk of scab for winter wheat, but not for spring wheat. Choosing the residue present option will usually indicate a higher risk than the no residue option. Finally, click on Indiana, or whatever state if of interest. The U.S. map will zoom in on a region. The map may display three different colors: salmon pink (high risk), yellow (moderate risk), or green (low risk). When I looked at the model prediction for a flowering date of 20 April, the risk for the entire region was low. The map also displays purple dots, which depict the location of hourly weather stations that provide data for the model. The model also uses a grid of virtual weather stations, which is the basis for the colors on the map. If a user clicks on weather stations closest to a field of interest, some graphs will appear below the map that show risk probabilities and weather data. The model does not allow prediction into the future because it uses real, hourly weather data, not predicted weather.
The model uses weather data for 7 days prior to wheat flowering. Research conducted in several states in the Upper Great Plains and the Corn Belt (including Indiana) indicates that weather during the week before flowering has a large impact on the amount of inoculum (spores) produced by Fusarium graminearum. The amount of inoculum in the air has a lot to do with the risk of head blight.
Model predictions can be improved if weather after flowering is also considered. However, a major goal of the model developers was to provide a decision aid to growers who are considering use of a fungicide to control the disease. Last year, Indiana and some other states had Section 18 exemptions for use of the fungicide Folicur (tebuconazole). Indiana has submitted a request for a Section 18 exemption for 2005, but has not yet received approval. When I learn of EPA’s decision, I will pass this information along in Pest&Crop.
A couple of cautionary notes: The model is not a perfect predictor of head blight. As stated on the web site, it has a prediction accuracy of about 80%. Last year, the model failed to predict the outbreak of head blight in Indiana. In my opinion, this was because the weather for the week before flowering over most of state was dry, and this lead to prediction of low risk. However, about the time wheat started flowering, rain started falling, and disease did develop. No model based on pre-flowering weather could have predicted this epidemic. These circumstances confirmed that wheat is susceptible to infection not just at the beginning of flowering, but throughout the flowering stages, and on into early grain development stages, something already known from inoculation studies in the greenhouse. Infection may occur as late as mid milk (GS 11.1). Post-flowering models could probably have predicted this epidemic, but the cut-off for applying Folicur is GS 10.51, so growers need a model that makes a prediction just before this stage. If this year we again have dry weather the week before flowering, growers may want to look at 7-day forecasts for the period just after flowering. If the prediction is for rain, a fungicide application may still be justified, despite a model prediction of low risk.
A second issue is that the model is based entirely on development of head blight symptoms in the field. These are seen as premature bleaching or all or a portion of the head. We know, however, that there is a poor correlation between head blight severity and symptoms in the grain (scabby kernels), and an even poorer correlation between head blight severity and levels of deoxynivalenol (DON) in grain. If head blight is severe, most likely the grain will be of poor quality and will contain DON. However, grain harvested from fields where head blight is not severe may also be scabby and contain high levels of DON. Grain appearance and DON levels are major concerns for millers and bakers, so scabby grain can be severely docked at the elevator. If testing reveals high levels of DON, a buyer may reject the grain entirely.
Despite these reservations, the model is a useful tool for management of Fusarium head blight. Information from the model, along with predicted weather can help make sound decisions about use of a fungicide.
![]()
Leaf and Head Blight of Wheat - (Gregory Shaner)
- A dry April portends little problem with Septoria blotch of wheat.
Wheat in Indiana is often infected by two leaf blight fungi: Septoria tritici and Stagonospora nodorum. Leaf blight is only a problem when rainfall is frequent during the growing season. The dry weather for most of April (notwithstanding the rain that started falling on the afternoon of the 20th) means that these diseases will not likely be a problem this year. This is especially so for Septoria leaf blotch. This disease requires frequent rainfall from late March through April. Several periods of 2 or more consecutive days with rain, when minimum daily temperatures remain above 47°F are the weather conditions that promote epidemics of Septoria blotch.
Stagonospora leaf blotch also requires frequent rains and warm weather to develop. It is favored by somewhat warmer weather than Septoria blotch. When rainy weather comes later in the season, as heads are emerging, it can develop rapidly. Should the weather during late April and May be much wetter than early April, Stagonospora leaf blotch could still develop.

Septoria tritici lesions on wheat leaf.
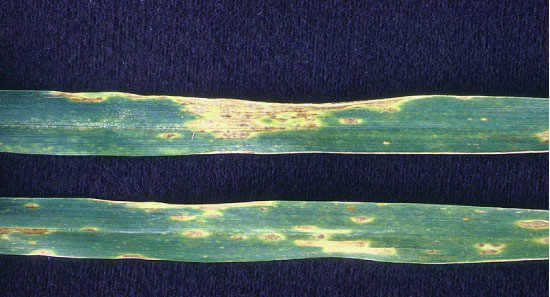
Stagonospora nodorum lesions on wheat leaves.
Symptoms of these two diseases are sometimes difficult to distinguish. The classical symptom of Septoria leaf blotch is a somewhat rectangular, tan lesion that contains numerous small, black specks. An early name for this disease was speckled leaf blotch, a name more descriptive and pronounceable than Septoria leaf blotch. The tiny block specks are fruiting bodies of the fungus—tiny, flask-shaped structures (pycnidia) embedded within the leaf and containing spores of the fungus. The classical symptom of Stagonospora leaf blotch is an elliptical, tan lesion with a dark spot in the center. The tan lesion is often surrounded by a yellow halo. I refer to these symptoms as classical because identification guides always show them. When these symptoms are present, a diagnosis can be made in the field with some confidence. However, leaves may show necrotic spots that aren’t clearly like one or the other of these symptoms. In this case, the only sure way to diagnose the disease is to examine lesions under a microscope.
Stagonospora nodorum is capable of infecting wheat heads as well as leaves. It produces a chocolate-brown discoloration on the glumes and lemmas. Like Septoria, this fungus also produces pycnidia, but in leaves they are difficult to see except with a microscope. However, the pycnidia of Stagonospora are quite obvious on glumes and lemmas, especially near the tips, as small, raised structures.
Wheat varieties differ in resistance to these pathogens. Some have a high degree of resistance to Septoria. There is no high degree of resistance to Stagonospora, but the rate of premature killing of upper leaves is slower on some varieties. The longer these upper two leaves remain green, the more time for grain filling and production of a quality crop.
![]()

