Pest & Crop Newsletter, Entomology Extension, Purdue University
- Eurpoean Corn borer Late But Not Out
- Nematode Updates
- Changes in SCN Soil Testing Policy
- Black Light Trap Catch Report
- Field Drydown of Mature Corn Grain
- Fear Monger Alert: Monitor Corn Fields for Weakened or Diseased Stalks
European Corn Borer Late But Not Out - (John Obermeyer, Christian Krupke, and Larry Bledsoe)
- Third brood corn borer flight is significant.
- Most borer larvae probably won't make it through the winter.
- Damage from this late flush will be minimal to most of Indiana's corn.
Recent black light catches (see “Black Light Trap Catch Report”) suggest that European corn borer is attempting a third generation; Indiana normally has two generations per year. This certainly comes as a surprise considering the low numbers of moths captured throughout the season.
This late season activity is likely a result of the hot summer we have had, but eggs laid now will likely have very poor survivorship, considering that at this late point in the season it is unlikely that most of the larvae will make it to the 5th instar, which is the overwintering stage. The last time we saw significant survival of a third generation was following the 1991 season, which was a tremendous corn borer year.
The only cornfields, or spots of fields, that will be attractive to egg-laying females are extremely late-planted/late pollinating fields, and corn that has yet to pollinate or is currently pollinating will essentially act as a “trap” crop by attracting the vast majority of corn borer moths in a given area. Infestations after the blister stage do not have as much of a physiological effect on yields as they do at earlier stages. However, the economics do not justify treating such late maturing corn. However, it is worthwhile to note that late-market sweet corn should be watched for this pest, as well as for the corn earworm.
![]()
Nematode Updates – (Jamal Faghihi and Virginia Ferris)
Jamal and Josh Hunnicutt made a follow-up to the July trip specifically to observe the soybean cyst nematode damage in White, Pulaski and Jasper cCunties, known to be the center of high SCN infestations, and to collect soil samples from fields with SCN symptoms. Soil processing in the laboratory is now under way. We knew that growers in these counties use SCN resistant cultivars extensively. We have had several reports by extension educators, and have observed ourselves that putative SCN resistant cultivars do not appear to be as resistant as they used to be. As you might know, the source of resistance for almost 97% of these cultivars is the same (PI 88788). We need to determine whether the breakdown in resistance is toward specific cultivars or whether the problem is related to the source of resistance for these cultivars. We collected soil samples from fields that we have followed for many years and for which we have maintained the original SCN populations on a susceptible cultivar in our greenhouse. A comparative study between present SCN populations and the original populations should give us a good indication as to whether any possible changes in nematode resistance might have occurred. We also randomly collected soil samples from fields that showed new SCN on the roots of soybeans. We will expose these SCN populations to PI 88788 to determine what percentage is likely to be susceptible. We will also subject all of the positive SCN soil samples that we receive this fall to PI 88788. Even though we might not know the exact nature of the early resistance to these SCN populations toward PI 88788, a combination of these on going and proposed studies should reveal to us whether changes have occurred. We will publish our findings as soon as the data become available.
If you have any questions about these or any other kinds of nematodes, you can contact Jamal Faghihi at 765-494-5901 or send an email to jamal@purdue.edu. Soil samples for nematode analysis can be sent to: Nematology Laboratory, Purdue University, Department of Entomology, Smith Hall, 901 W. State Street, West Lafayette, IN 47907-2089.
![]()
Changes in SCN Soil Testing Policy– (Jamal Faghihi and Virginia Ferris)

Close-up symptoms of SCN on soybean leaves.

SCN symptoms in an infested soybean field, notice circle of yellow stunted beans.

Symptoms of SCN in a field planted with a resistant cultivar.
We were just informed about a change in policy by the Indiana Soybean Board in regard to the SCN soil testing program. As you may know in the past the Indiana Soybean Board has paid the SCN soil processing fee for up to 10 samples/grower/year. The Indiana Soybean Board will continue to pay this fee through the end of December 2005. Samples submitted on or after January 1, 2006 will no longer be the financial responsibility of the Indiana Soybean Board, but the responsibility of the submitter.
We thought it is important to inform you about this change as soon as possible. Sampling for SCN is crucial for sound management of the soybean cyst nematode. We hope that growers will continue to monitor their SCN populations by means of soil sampling and that this change in ISB policy does not have a severe impact on their decisions.
If you have any questions about these or any other kinds of nematodes, you can contact Jamal Faghihi at 765-494-5901 or send an email to jamal@purdue.edu. Soil samples for nematode analysis can be sent to: Nematology laboratory, Purdue University, Department of Entomology, Smith Hall, 901 W. State Street, West Lafayette, IN 47907-2089.
Bug Scout

![]()
Click for Table.
Black Light Catch Report
![]()
Soybean Rust Update for Indiana – (Greg Shaner)
Observations
No Asian soybean rust has been found in Indiana. Sentinel plots are being scouted regularly. Bacterial pustule, brown spot, downy mildew, and bacterial blight have been found in some fields.
Growth Stages
Many fields are now in the seed development growth stages (R5 to R6). Early in the season, plants in many fields seemed shorter than normal, but where rains have fallen, plants are of normal height (36 to 42 in.) and canopies are thick.
Management
There is no reason at this time to spray a fungicide on soybean for rust control. Although there have been several new findings of rust in sentinel plots and a few fields in the South, the disease has not been found north of about 33 degrees latitude. Unless there is a sudden and dramatic change in the rate of spread of the rust fungus, it is unlikely to pose any threat to Indiana’s soybean crop this year.
Forecast Outlook
Weather conditions in Indiana are seemingly favorable for rust, but the slow rate of spread and generally low severity of rust in the South, plus the distance of known sources of rust from Indiana, make it unlikely that rust will reach here while the crop is still vulnerable, if rust arrives at all.
Scouting Recommendations
Although the risk of damage from rust is now low, growers are encouraged to continue scouting fields so that if rust does reach Indiana this can be documented.
Scouting Techniques
The goal of scouting is to detect rust at a very low incidence (fewer than 5% of plants with any visible infection), when effective control with a fungicide is still possible. At least 150 leaves should be carefully examined. If initial spore loads are light, there may be only a few infections per acre. When these infections mature and produce spore-bearing pustules, which takes about 7-9 days, most of those spores will land near where they were produced. Thus, initial disease development will be focal. That is, there will be a few “hot spots” in a field where rust is severe, with little or no rust outside these hot spots. These hot spots, or foci, may be only 1 yard in diameter. As a focus develops and more spores are produced, infection in the field will become more general. However, the goal of scouting is detect rust before it becomes general. To maximize the chances of finding a focus, it is better to look at many areas in a field, but only a few plants in each area, rather than to look at a lot of plants in only a few areas. If there are areas in a field where leaves tend to be wet longer (low areas, areas shaded in the morning by woods, etc.), scouting should concentrate on these. Examine the underside of leaves because this is where pustules preferentially develop. Look in the lower to mid canopy for rust, on the third or fourth leaf below the top fully developed leaf. The Purdue Plant and Pest Diagnostic Laboratory web site has images and tips for recognizing soybean rust and distinguishing it from other foliar diseases. See www.ppdl.purdue.edu/ppdl/pubs/soybean_rust_symptoms_web.pdf.![]()
Frogeye Leaf Spot on Soybean - (Gregory Shaner and Andreas Westphal)
- A "southern" disease of soybean can be found in many norhtern Indiana soybean fields.
In Issue 19 of Pest&Crop (July 29) we reported that frogeye leaf spot was showing up in several Indiana soybean fields, farther north than customarily seen in Indiana. We continue to see it in northern Indiana, and have received many reports of the disease from others in this part of the state.
The earlier article discusses symptoms of the disease, which are quite distinct from other leaf diseases likely to be encountered in Indiana.
Frogeye leaf spot is by no means widespread. The disease is found only in some varieties. Many varieties are evidently resistant.
![]()
White Mold of Soybean – (Gregory Shaner, Andreas Westphal)
- White mold is starting to show up in some northern Indiana fields.

Figure 1. Plant with white mold. White areas on lower stems are points of infection, covered with mycelium of the fungus. Leaves are wilted, but still attached to the plant.

Figure 2. White mold lesion on lower stem. Infected area is bleached out.

Figure 3. Sclerotia of Sclerotinia sclerotiorum.
White mold (Sclerotinia stem rot) is appearing in some northern Indiana soybean fields. This is later than normal for this disease. Sudden death syndrome is widespread in northern Indiana this year, so patches of dying plants should be inspected to determine the cause.
White mold is first evident as scattered wilted plants among healthy plants. Leaves of infected plants wilt, but remain attached to the stem (Fig. 1). The soybean stem will have bleached lesions 1 to 5 inches long, often centered on lower nodes (Fig. 2). These lesions are often covered with cottony mycelium of the fungus. Embedded within this mycelium are sclerotia--hard, black structures of 0.1 to 1 inch long (Fig. 3). Greater numbers of sclerotia may be found within the stem. These sclerotia can survive in soil for several years. They are also an important means of dispersal.
Yield reduction depends on when white mold first affects a soybean plant and how many plants in a field are infected. A girdling stem lesion largely prevents movement of water and nutrients from the root system to the foliage. If the soybean has not matured by the time a girdling stem lesion develops, seeds may remain small and be lost during harvest. Seed of plants affected later may be of more normal size.
A fungus known as Sclerotinia sclerotiorum causes white mold. The pathogen may be introduced into previously non-infested fields with soybean seed contaminated with sclerotia. Sclerotia are similar to soybean seed in size and density, and can be mixed with seed from infested fields when harvesters do not separate them from the seed. Modern seed cleaning equipment does a good job of removing sclerotia. The fungus can be spread within a field during harvest, when sclerotia are blown out the back of the combine with chaff.
An infested field needs special management considerations. If there are only a few affected areas in a field, it might be better to not harvest these, to avoid dispersing sclerotia. The number of viable sclerotia in a field will decline over time if there are no susceptible hosts in a field. Corn and small grains are not hosts for this fungus, but many broadleaf weeds can be infected. These include pigweeds, ragweeds, and several mustards. Therefore, good weed control is necessary, regardless of the crop in a field. Many vegetable crops are also susceptible, which may also be why the disease is more common on soybean in northern Indiana, where more vegetables are produced. Wider row spacing (30 inches) may reduce severity of white mold. This is because at the time flowering begins, the canopy has not closed, so microclimate conditions at the soil surface may be less conducive for production of spores and for infection. A grower should choose a less susceptible soybean cultivar for production in high-risk fields.
![]()
Soybean Sudden Death Syndrome – (Andreas Westphal, Scott Abney, and Gregory Shaner)
- Drought stress followed by heavy rains is followed by widespread SDS.

Figure 1. Soybean field with severe SDS in foreground. Patches of SDS can be seen in the distance. It is typical to see areas of severe disease interspered with healthier plants.

Figure 2. Interveinal chlorosis and necrosis on a leaflet from a plant with SDS. Shortly after this stage of symptom development, leaflet will shrivel and drop from the petiole.

Figure 3. An excellent diagnostic symptom of SDS can be seen in the split lower stem.There are brown streaks in the cortex, but the pith is a normal color.
A few weeks ago (Pest&Crop, July 22, 2005, Issue 18) we cautioned that SDS could develop in many areas of Indiana following the midsummer rains. This disease is caused by the soil-borne fungus Fusarium solani f. sp. glycines with the newly proposed name Fusarium virguliforme. Early infection/colonization of young soybean seedlings frequently occurs in cool and moist soils soon after planting. These conditions are likely when soybean is planted early. High soil moisture triggers the development of foliar symptoms of SDS. If soils are wet at the beginning of the reproductive growth stages, foliar symptoms of SDS may appear any time from mid July through mid August.
During most of the early summer, heat and drought caused stress in many areas of Indiana. The soybean cyst nematode caused significant damage that was easily detectable. These stressed crops then received rains from late July through early August. While this probably rarely caused soil moisture saturation, soil water content was apparently sufficient in many areas to trigger the development of SDS. Similar to previous years, the distribution of SDS probably is more a reflection of weather patterns than of the distribution of the pathogen in fields over the state. Although SDS can be found in some fields in southern Indiana, the disease is not nearly as widespread there as it was in 2004. This year, SDS is more widespread in northern Indiana. Symptoms are fairly uniform throughout some fields, rather than in patchy patterns typically seen in previous years. This may be the result of a uniform stress pattern of drought followed by overabundance of moisture. It also demonstrates that in many fields, the pathogen is widely distributed.
There is a clear association between delayed planting and reduced severity of SDS. An extreme delay will reduce yield potential because the plant does not have sufficient vegetative growth before flowering. There needs to be a compromise when making planting time decisions, between reducing the risk of SDS and losing yield potential from planting too late.
While soybean varieties with some resistance to SDS are slowly replacing the most susceptible ones, it remains important to not plant too early in fields with a history of SDS. Good note keeping of which fields show SDS in 2005 will aid planting decisions in 2007 when many 2005 soybean fields will once again be planted to soybean. Fields with severe SDS in 2005 should probably be the last ones planted to soybean in 2007. This will not eliminate SDS but has potential to reduce the risk of severe damage. It is also important to select a variety with the best possible resistance to SDS, in combination with other required traits.
![]()
Field Drydown of Mature Corn Grain – (Bob Nielsen)
- Weather conditions are the major influence on in-field grain drydown.
- Plant characteristics can also influence in-field grain drydown.
- Early grain maturation usually means faster in-field grain drydown.
- Later grain maturation usually means slower in-field grain drydown.
As Indiana’s corn crop heads into the last half of the grain filling period, growers’ thoughts begin to focus on the upcoming harvest season. As of 14 August, Indiana’s corn crop is developing at roughly an average pace with approximately 76% of the state’s crop at or beyond the dough stage of kernel development (USDA-NASS, 15 Aug 2005). If this pace continues, half of the state’s crop should be mature by mid-September; again about an average date.
Grain moisture content at harvest obviously influences growers’ cost of artificially drying the grain after harvest. An early drydown of the crop also facilitates early or at least timely harvest of the crop prior to the colder and, often, wetter conditions of late fall.
Kernel moisture content decreases as the kernel develops through the blister stage (~ 85% moisture), milk stage (~ 80% moisture), dough stage (~ 70% moisture), dent stage (~ 55% moisture), and finally physiological maturity (~ 30% moisture). Prior to physiological maturity, decreases in kernel moisture occur from a combination of actual water loss (evaporation) from the kernel plus the continued accumulation of kernel dry matter via the grain filling process. After physiological maturity, percent kernel moisture continues to decrease primarily due to water loss from the kernel.
Weather & Timing of Grain Maturation
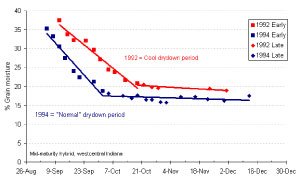
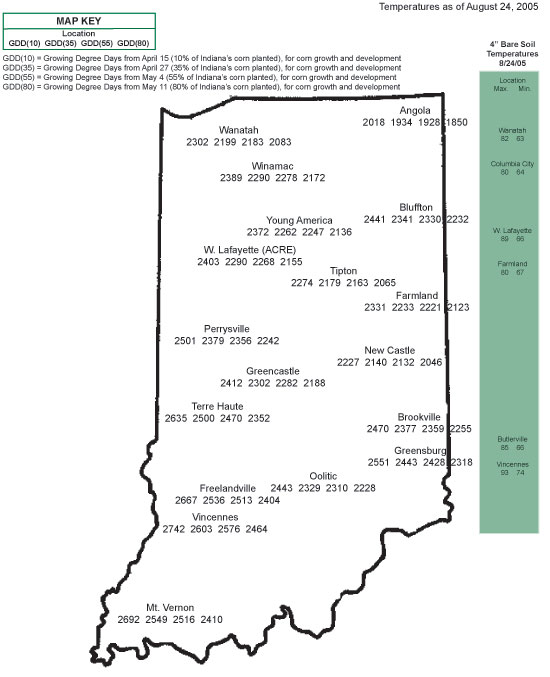
Figure 1. Decrease in grain moisture content of a mid-maturity corn hybrid over time (westcentral Indiana).
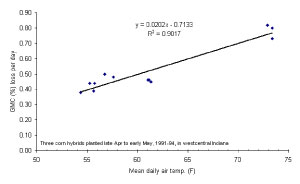
Figure 2. Average daiily grain moisture loss (percentage points/day) relative to average daily air temperatuer during the drydown period for three corn hybrids planted late April to early May, 1991-1994 (westcentral Indiana).
Figure 3. Relationship between daily grain moisture loss (drydown) and the date at which the grain nears maturity (half-milking) for three corn hybrids planted late April to early May, 1991-1994 (westcentral Indiana).
Grain moisture loss in the field occurs at a fairly linear rate within a range of grain moisture content from about 40 percent down to 15 to 20 percent, and then tapers off to little or no additional moisture loss after that. Figure 1 illustrates changes in grain moisture content over time for an adapted medium maturity hybrid grown in Indiana in 1992 (unusually cool fall) and 1994 (more typical fall temperatures).
Field drying of mature corn grain is influenced primarily by weather factors (especially temperature and humidity) and, to a somewhat lesser degree, by ear and husk characteristics of hybrids. Simply put, warmer temperatures and lower humidity encourage rapid field drying of corn grain (Fig. 2).
Because grain drydown rates are greater when the drydown period is warmer, it stands to reason that a corn crop that matures in late August will dry down faster than one that matures in mid-September. In fact, there is a close relationship between the date when the grain nears physiological maturity (half-milkline or 2-3 weeks prior to kernel blacklayer) and the subsequent average daily drydown rate. Average daily drydown rates will range from about 0.8 percentage point per day for grain that nears maturity in late August to about 0.4 percentage point per day for grain that nears maturity in mid- to late September (Fig 3).
Bear in mind that grain moisture loss for any particular day may be quite high or low depending on the exact temperature, humidity, sunshine, or rain conditions that day. It is not unheard of for grain moisture to decline more than one percentage point per day for a period of days when conditions are warm, sunny and dry. By the same token, there may be zero drydown on cool, rainy days.
Hybrid Variability for Field Drying
Hybrid variability for the rate of grain moisture loss during post-maturity drydown and the eventual grain moisture content at harvest are of great interest to grower and seed industry alike. Growers desire hybrids with superior yielding ability (maximum gross income) that also dry very quickly in the fall (minimum drying or grain shrinkage costs).
The seed industry uses grain moisture content data to assign relative hybrid maturity ratings on the basis of relative moisture differences among hybrids at harvest. Two hybrids that differ in one “day” of relative maturity will typically vary by about one half percentage point of grain moisture content (an average daily loss of moisture) if planted and harvested on the same days. Recognize that relative hybrid maturity ratings are most consistent within, not among, seed companies.
When weather conditions are not favorable for rapid grain drydown, then hybrid characteristics that influence the rate of grain drying become more important. Researchers have identified the following traits or characteristics as ones most likely to influence grain drying in the field. The relative importance of each trait varies throughout the duration of the field drydown process and, as mentioned earlier, is most influential when weather conditions are not conducive for rapid grain drying.
- Husk Leaf Number. The fewer the number of husk leaves, the more rapid the grain moisture loss. In fact, modern hybrids have fewer husk leaves than those commonly grown years ago.
- Husk Leaf Thickness. The thinner the husk leaves, the more rapid the grain moisture loss.
- Husk Leaf Senescence. The sooner the husk leaves senesce (die), the more rapid the grain moisture loss.
- Husk Coverage of the Ear. The less the husk covers the tip of the ear, the more rapid the grain moisture loss.
- Husk Tightness. The looser the husk covers the ear, the more rapid the grain moisture loss.
- Ear Declination. The sooner the ears drop from an upright position after grain maturation to a downward position, the more rapid the grain moisture loss. In particular, husks of upright ears can “capture” rainfall.
- Kernel Pericarp Characteristics. The pericarp is the outermost layer of a corn kernel (botanically; the ovary wall). Thinner or simply more permeable pericarp layers have been associated with faster drying rates in the field.
Final Trivia For Coffeeshop Conversations
Post-maturity grain moisture loss occurs primarily by evaporative loss from the kernel itself. Research many years ago established that post-maturity moisture loss through the kernel connective tissues (placental tissues) back to the cob is essentially non-existent (Kiesselbach and Walker, 1952). As those tissues cease to function (associated with the onset of physiological maturity), the moisture and nutritional connection between kernel and cob is essentially broken.
Related References
Cavalieri, A.J. and O.S. Smith. 1985. Grain Filling and Field Drying of a Set of Maize Hybrids Released From 1930 to 1982. Crop Sci. 25:856-860.
Crane, P.L., S.R. Miles, and J.E. Newman. 1959. Factors Associated With Varietal Differences in Rate of Field Drying in Corn. Agron. J. 51:318-320.
Kiesselbach, T. A. and Elda R. Walker. 1952. Structure of Certain Specialized Tissue in the Kernel of Corn. Am J Bot 39:561-69
Nielsen, R.L. (Bob). 2004. Grain Fill Stages in Corn. Corny News Network, Purdue Univ. Online at www.kingcorn.org/news/articles.04/GrainFill-0705.html [URL verified 8/8/05].
Troyer, A.F. and W.B. Ambrose. 1971. Plant Characteristics Affecting Field Drying Rate of Ear Corn. Crop Sci. 11:529-531.
USDA-NASS. 15 Aug 2005. Indiana Crop & Weather Report. U.S. Dept. Ag - Nat’l Ag. Statistics Service. Online at www.nass.usda.gov/in/cropweat/2005/we3305.pdf [URL verified 8/15/05].
![]()
Fear Monger Alert: Monitor Corn Fields for Weakened or Diseased Stalks - (Bob Nielsen)
During the grain filling period, developing kernels become a significant photosynthetic “sink” for the products of photosynthesis and respiration. Corn plants prioritize the movement of these photosynthates to the kernels, even at the expense of not maintaining cellular health of stalk, leaf, and root tissues.
The primary effect of severe stress on a corn plant (drought, heat, nutrient deficiency, leaf diseases, insect damage, hail damage, consecutive days of cloudy weather) is a reduction in photosynthetic rates. If photosynthetic capacity decreases significantly during grain fill, plants often respond by remobilizing stored carbohydrates from stalk and leaf tissues to supply the intense physiological demand by the developing grain on the ears. In addition to physically weakening the stalk of plants, remobilization of stored carbohydrates and/or the consequent lower cellular maintenance of root and stalk tissues increases the susceptibility of the plant to root and stalk rots.
Reports have already begun to trickle in from several areas of Indiana about weak plants with varying degrees of root and stalk rot development. Growers should monitor stressed fields the remainder of this month and into early September for compromised stalk strength or the development of severe stalk rots and adjust their harvest schedules accordingly to harvest these fields early in the season before that one big storm brings the crop to its knees.
Related References
Shaner, G. and D. Scott. 1998. Stalk Rots of Corn. Purdue Univ. Extension Publication BP-59. Available online at www.ces.purdue.edu/extmedia/BP/BP-59.pm65.pdf (URL verified 8/23/05).
Vincelli, Paul. 2004. Factors That Could Enhance Stalk Rots in Corn. Kentucky Pest News (Aug 2). Univ. of Kentucky. Available online at www.uky.edu/Agriculture/kpn/kpn_04/pn040802.htm#corrot (URL verified 8/23/05).
For other Corny News Network articles, browse through the CNN Archives at www.kingcorn.org/news/archive.html. For other information about corn, take a look at the Corn Growers’ Guidebook at www.kingcorn.org.
![]()
Indiana and Ohio CCA Program at the Farm Science Review – (Harold Watters, Ohio State University)
Attending the Farm Science Review is an old tradition in Ohio. This year we want to encourage you to attend the Farm Science Review to do more than just see large equipment and talk with seed sales representatives. Ohio State University in cooperation with Purdue University will be presenting a Certified Crop Adviser program at the Review, to include crop updates from the growing season and hands on participation in evaluating applied treatments. We will have a very good update on Asian soybean rust from two state authorities. And with the price of nitrogen fertilizer increasing and the concern for water quality, we will also be able to discuss management practices to better manage this important crop input.
Program:
Soybean Rust: Indiana & Ohio Respond and A Review of 2005
- Anne Dorrance, OSU Plant Pathologist
- Greg Shaner, Purdue Plant Pathologist
Nutrient Management: Efficiency and Water Quality
- Robert Mullen, OSU Soil Fertility Specialist
- Nathan Watermeier, OSU Extension Technology Lead
Manure Application: Water Quality, Needs and Efficiency
- Brad Joern, Purdue Agronomist
- Jon Rausch, OSU Ag Engineering
Diagnosing Kernel Set & Grain Fill Problems in Corn
- Peter Thomison, OSU Corn Specialist
- Bob Nielsen, Purdue Corn Specialist
We are planning a program to start at 8AM on Wednesday of the Farm Science Review, September 21st, and end at noon with a lunch. Wednesday is the big day for attendance so we thought providing a lunch would help you with planning your activities for the rest of the day at the Review. During lunch you will have time for interaction with other CCAs and time to ask questions of the state specialists. After lunch you can spend the rest of the day making those important visits to the field demonstrations or talking with on-site exhibitors.
For the CCA continuing education credits, meal, a parking pass, a ticket to the Farm Science Review and access to some of Indiana and Ohio’s best state specialists, we will charge $70. Registration is limited to 120, please register by September 7th. To register click on “CCA College” at the OSU Extension Agronomic Crops Team Website - http://agcrops.osu.edu/, on the Purdue Diagnostic website - www.agry.purdue.edu/DTC/ or by calling (419) 466-4145 (T&M Registration Services).
![]()