
Pest & Crop
Newsletter
Purdue Cooperative Extension Service

Purdue Cooperative Extension Service
Issue 1, March 20, 2015 • USDA-NIFA Extension IPM Grant
CLICK HERE FOR A PDF VERSION OF THIS ISSUE ![]()
Plant parasitic nematodes are microscopic worm-like organisms that require water to survive and are sensitive to high temperature. Only living nematodes can be extracted from roots. Through the years we have had many samples submitted to the Purdue Nematology Laboratory with little consistency in the quality of the samples. We have discussed proper sampling procedures at every opportunity but not exclusively until now. Sampling might appear trivial but we believe proper sampling is the most crucial step for correct diagnosis. Because we continue to receive improper samples we address this issue again via this article. Even though procedures for sampling among the most important plant parasitic nematodes are similar, there are differences based on the host and the type of nematodes we are trying to recover.

Corn parasitic nematodes: There are three major groups of nematodes that parasitize corn.
Endo-parasites (e.g., Lesion nematodes): These nematodes mostly feed within corn roots. Plant roots along with surrounding soil must be submitted to recover these types of nematodes. A proper soil sample consists of about one quart of sub-samples taken to a depth of 6-8 inches directly from the root zone of affected corn plants Dig up the stunted plants and place with adhering soil and roots in a plastic bag. Attach a label to the outside of the bag. On the label, give sufficient information to identify the sample. Root and soil samples should not become dry or be exposed to high temperature. The best time to sample for these nematodes is mid-season when most of the nematodes have migrated to the inside of the roots. These nematodes continue to feed throughout the growing season. They can be found in all kinds of soil types.
Ecto-parasites (e.g., Needle nematodes): These types of nematodes feed from outside of the young roots. The sampling procedure is the same as described above for Lesion nematodes. But, Needle nematode is mostly a problem in sandy soil and can be found early in the season (4-6 weeks after germination). Often they disappear when the soil temperature rises above 80 degrees.
Semi endo-parasites (e.g., Lance nematode): These nematodes can feed from inside or outside of the roots. The sampling procedure is the same as that described above for endo-parasites. Lance nematodes feed throughout the season, have no soil type preference and can parasitize corn or soybean.
Soybean parasitic nematodes: Lesion and Lance nematodes parasitize soybean too but Needle nematode does not. The sampling procedure for these nematodes in soybean is similar to the one described for corn. The most economically important nematode affecting soybean is the Soybean Cyst Nematode (SCN). The SCN distribution, as for most of the plant parasitic nematodes, is in patches. So it is very important to take many sub-samples to increase possibility of hitting the concentrated areas. A soil probe or a small trawl should be used to collect the sub-samples. Most of these nematodes are within 6-8 inches of the soil. One sample for every 10 acres is ideal. A quart of soil is sufficient and no root samples are required for SCN. Samples can be taken anytime.
| Recommended optimum sampling type and time for major plant parasitic nematodes in Indiana. |
|---|
| Host | Target nematode | Sample type | Optimum time to sample |
|---|---|---|---|
| Corn | Needle | Soil and roots | June-mid July |
| Corn | Lesion, Lance | Soil and roots | Late June-Late August |
| Soybean | Lesion, Lance | Soil and roots | Late June-Late August |
| Soybean | SCN | Soil | Anytime |
| Turf | All | Soil and roots | June and September |
| Melons | Root knot | Roots | At harvest |
| Mint | Lesion | Roots and soil | Late June-September |
| Mint | Needle | Roots and soil | Late Spring or early fall |
| Mint | Root knot | Roots | Fall |
The Purdue Weed Science team has created Two time-lapse videos using pictures taken by cameras placed at their palmer amaranth research site last growing season. The early growth video captures the emergence of the soybean crop and Palmer amaranth from early May through early June. The video emphasizes the utility of pre-emergence herbicides by capturing treated vs non-treated plots.
A late growth video captured from the same site shows the rapid growth of Palmer amaranth during the month of July 2014.
Current efforts are underway to continue to produce time lapse video’s capturing Palmer amaranth growth as well as plans to place more plot cameras on field research this upcoming summer.
Purdue Weed Science has also produced the following publications and videos pertaining to Palmer amaranth.
Palmer Amaranth Biology, Identification, and Management: https://mdc.itap.purdue.edu/item.asp?Item_Number=WS-51#.VQG-SmY0rbp
Palmer amaranth Seedling ID (Video):
Identifying Palmer Amaranth in The Field:
Appropriate nitrogen (N) management for corn is important for maximizing profit and minimizing environmental impact. For best results N fertilizer should be applied shortly before or after corn planting up to about the V7 growth stage. Anhydrous ammonia and/or urea ammonium nitrate (UAN – 28-32%) N fertilizers when injected or incorporated are generally the most consistently effective and efficient. Urea containing fertilizers left on the soil surface can lose ammonia to the atmosphere especially when broadcast on crop residues.
Choosing the correct rate of N fertilization in any particular year is difficult because soil, weather, and crop interactions can result in a different optimum rate for all combinations of these factors. Since the weather is impossible to predict usually the best we can do is suggest a research-based N rate guideline that is likely to produce the highest profit over a number of years.
To obtain our N rate guidelines we conducted more than a hundred field scale N response trials on Purdue farms and farmer’s fields beginning in 2006. Efficient timing, sources, and placement of N were used to identify the minimum N rate needed to maximize yield (AONR-agronomic optimum N rate) given the N loss conditions encountered each year. Results were aggregated for regions of the state and economic optimum N rates (EONR) were calculated across a range of N and grain values.
Guidelines are for corn grown in rotation with soybean on medium- and fine-textured soils are shown for regions of Indiana in Figure 1. Corn grown after corn requires 40-50 pounds of N per acre more than corn after soybean.
For more detailed information see: Nitrogen Management Guidelines for Corn in Indiana.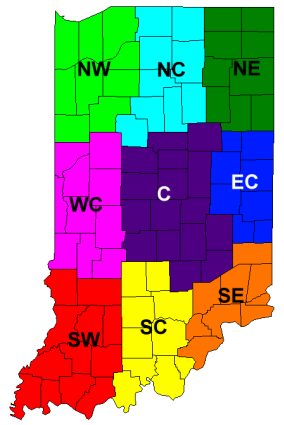
| Figure 1. Agronomic (AONR) and economic (EONR) optimum N rate guidelines for corn grown in rotation with soybean for medium- and fine-textured soils for different regions of Indiana and for sandy soils across Indiana. EONR was calculated with $4/bushel corn and $0.59/pound of N ($960/ton anhydrous ammonia or $330/ton urea ammonium nitrate). |
|---|
| Map region | AONR | EONR |
|---|---|---|
| SW & WC | 183 | 158 |
| NW & NC | 191 | 166 |
| NE | 226 | 191 |
| EC | 233 | 201 |
| SE | 196 | 171 |
| C | 221 | 188 |
| Sandy soils | 184 | 162 |
| SC | Insufficient data | Insufficient data |
The 2015 Popcorn Agri-Chemical Handbook is now available to ensure everyone in the popcorn industry is informed about products registered for use on popcorn or in popcorn storage facilities. The handbook lists agri-chemicals registered and the regulatory status or special use restrictions, if any.
The handbook continues to provide appendix information on residue tolerances, as may be found in the Global MRL Database, which includes popcorn (corn, pop) and denotes established levels by the US, Codex, EU and 88 markets.
The handbook notes the Mode or Mechanism of Action (MOA) numerical classification of each listed chemical when used on a product label. The classification schemes are published by the Insecticide Resistance Action Committee, the Herbicide Resistance Action Committee and the Fungicide Resistance Action Committee. The handbook also highlights the Signal Word “Danger” when used on a product label as required by the EPA’s Label Review Manual.
The Popcorn Board urges you to provide the above links to growers or download, print and distribute the updated version of this critical information to them. Contact Genny Bertalmio, +1.312.821.0217 or gbertalmio@smithbucklin.com, for further information.
The Popcorn Board accepts voluntary contributions to ensure continued funding of its efforts to provide this important information to the popcorn industry. Checks should be mailed to The Popcorn Board, 8333 Solutions Center, Chicago, IL 60677-8003.| Entomology Dept. Ext. Web Site: extension.entm.purdue.edu |
|||
|---|---|---|---|
| Yaninek, Steve | (765) 494-4554 | yaninek@purdue.edu | Head, Dept. of Entomology |
| Bledsoe, Larry | (765) 494-8324 | lbledsoe@purdue.edu | Field Research, CAPS |
| Faghihi, Jamal | (765) 494-5901 | jamal@purdue.edu | Nematology |
| Hunt, Greg | (765) 494-4605 | hunt@purdue.edu | Beekeeping |
| Krupke, Christian | (765) 494-4912 | ckrupke@purdue.edu | Field Crop Insects |
| Loven, Judy | (765) 494-8721 | loven@purdue.edu | USDA, APHIS, Animal Damage |
| Mason, Linda J. | (765) 494-4586 | lmason@purdue.edu | Food Pest Mgmt. & Stored Grain |
| Obermeyer, John L. | (765) 494-4563 | obe@purdue.edu | Field Crops Insects & IPM Specialist |
| Tammy Luck | (765) 494-8761 FAX: (765) 494-7197 |
luck@purdue.edu | Administrative Assistant |
| Agronomy Dept. Ext. Web Site: ag.purdue.edu/agry/extension |
|||
| Anderson, Joe | (765) 494-4774 | janderson@purdue.edu | Head, Dept. of Agronomy |
| Brouder, Sylvie | (765) 496-1489 | sbrouder@purdue.edu | Plant Nutrition, Soil Fertility, Water Quality |
| Camberato, Jim | (765) 496-9338 | jcambera@purdue.edu | Soil Fertility |
| Casteel, Shaun | (765) 494-0895 | scasteel@purdue.edu | Soybean and Small Grains Specialist |
| Gerber, Corey | (765) 496-3755 | gerberc@purdue.edu | Director, Diagnostic Training Center |
| Joern, Brad | (765) 494-9767 | bjoern@purdue.edu | Soil Fertility, Nutrient Management |
| Johnson, Keith D. | (765) 494-4800 | johnsonk@purdue.edu | Forages |
| Mansfield, Charles | (812) 888-4311 | cmansfie@purdue.edu | Small Grains, Soybean, Corn (SWIN) |
| Nielsen, Robert L. | (765) 494-4802 | rnielsen@purdue.edu | Corn, On-farm Research, Precision Agriculture |
| Steinhardt, Gary | (765) 494-8063 | gsteinha@purdue.edu | Soil Management, Tillage, Land Use |
| Vyn, Tony | (765) 496-3757 | tvyn@purdue.edu | Cropping Systems & Tillage |
| West, Terry | (765) 494-4799 | twest@purdue.edu | Soil Management & Tillage |
| Lisa Green | (765) 494-4783 FAX: (765) 496-2926 |
lgreen06@purdue.edu | Extension Secretary |
| Botany and Plant Pathology Dept. Ext. Web Site: www.ag.purdue.edu/btny/Extension |
|||
| Goldsbrough, Peter | (765) 494-4615 | goldsbrough@purdue.edu | Head, Dept. of Botany & Plant Pathology |
| Creswell, Tom | (765) 494-8081 | creswell@purdue.edu | Director Plant & Pest Diagnostic Laboratory |
| Johnson, Bill | (765) 494-4656 | wgj@purdue.edu | Weed Science |
| Legleiter, Travis | (765) 496-2121 | tlegleit@purdue.edu | Weed Science |
| Ruhl, Gail | (765) 494-4641 | ruhlg@purdue.edu | Plant & Pest Diagnostic Laboratory |
| Whitford, Fred | (765) 494-4566 | fwhitford@purdue.edu | Purdue Pesticide Programs |
| Wise, Kiersten | (765) 496-2170 | kawise@purdue.edu | Field Crop Diseases |
| Woloshuk, Charles | (765) 494-3450 | woloshuk@purdue.edu | Mycotoxins in Corn |
| Lisa Gross | (765) 494-9871 FAX: (765) 494-0363 |
gross25@purdue.edu | Extension Assist./P&PDL Lab Coordinator |
| Agricultural & Biological Engineering Dept. Ext. Web Site: engineering.purdue.edu/ABE |
|||
| Engel, Bernie | (765) 494-1162 | engelb@purdue.edu | Head, Dept. of Ag. & Bio. Engineering |
| Frankenberger, Jane | (765) 494-1194 | frankenb@purdue.edu | GIS and Water Quality |
| Kelley, Lyndon | (269) 467-5511 | kelleyl@msu.edu | Irrigation Educator |
| Ileleji, Klein | (765) 494-1198 | ileleji@purdue.edu | Post Harvest Grain Quality |
| Ni, Jiqin | (875) 496-1733 | jiqin@purdue.edu | Manure Management |
| Carol Weaver | (765) 494-1174 F: (765) 496-1356 |
sikler@purdue.edu | Extension Assistant |
Purdue Extension Entomology
901 W. State Street
West Lafayette, IN, 47907
(765) 494-8761
luck@purdue.edu
@PurdueExtEnt
PurdueEntomology

If you would like to be alerted by e-mail when the current issue of the Pest&Crop is available on-line, please enter your e-mail address and click the submit button.
It is the policy of the Purdue University Cooperative Extension Service that all persons have equal opportunity and access to its educational programs, services, activities, and facilities without regard to race, religion, color, sex, age, national origin or ancestry, marital status, parental status, sexual orientation, disability or status as a veteran. Purdue University is an Affirmative Action institution. This material may be available in alternative formats.