Pest & Crop Newsletter
|
|||||||||
A Follow-up on Nematodes Damaging corn and Soybean- (Jamal Faghihi and Virginia Ferris) As we predicted back in May , this was a bad year for Needle Nematode in corn. The good news is that the damage is done and we no longer find this nematode. When the soil temperature rises, this nematode simply disappears and we are no longer able to extract it from the soil. Damaged corn, for most part, will start to recover, even though some yield loss should be expected. The story for soybean is completely opposite. The Soybean Cyst Nematodes (SCN) are thriving and the first generation of this nematode has matured and the second generation is underway. This time of the year is the best time to observe the white and yellow SCN females on the roots. The best way to observe this nematode is to dig the whole plant root system with the soil ball around it. Dip the soil ball into a bucket of water and gently shake the soil away from the roots. The white or yellow SCN females should be visible on the roots and can be observed with the naked eye. They are the size of a grain of sugar. Recognition of this nematode in a timely fashion is extremely important. The yellow stunted patches of soybean, a typical SCN symptom, might not always be present in the soybean field although the damaging nematodes are present. Thus, we recommend close observation of soybean roots by every soybean grower despite lack of the presence of typical symptoms. The Indiana Soybean Board is continuing to cover the cost of soil analysis for SCN (up to 10 samples/grower/year). If you have questions about these, or any other kinds of nematodes, you can call us at 765-494-4611. If you are uncertain about whether plant parasitic nematodes are the cause of some type of unsatisfactory crop growth, you might consider sending a soil sample to us at: Nematology Laboratory, 1158 Smith Hall, Department of Entomology, Purdue University, West Lafayette, IN 47907-1158.
Rain and Spider Mites– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
Scattered thunderstorms have brought moisture relief to some parched areas of Indiana. Now the question concerning spider mites is whether the rain will “control” them. Before attempting to answer the question, let’s review the factors that come together to create a spider mite problem in soybean fields.
Extended hot and dry conditions will:
A significant rainfall (1 inch or more) followed by high humidity will:
Remember, rain does not make the spider mites go away! Unless fungal pathogens wipe them out (given continual high humidity), they will lurk and possibly remerge should the weather return to hot and dry. Also, spider mite damage is irreversible. That is, plants that are severely bronzed or brown will not green back up and contribute to the yield of the field. Refer to last week’s Pest&Crop for control materials and application techniques.
Monitoring and Decision Rules for Western Corn Rootworm Beetles in Soybean– (John Obermeyer, Larry Bledsoe, Rich Edwards, and Corey Gerber) The following is from Extension Publication E-218. A color PDF version of this publication can be downloaded at: <http://www.entm.purdue.edu/entomology/ext/targets/e-series/fieldcro.htm>. Why is there rootworm damage in corn following soybean? What can be done to reduce unnecessary insecticide applications? How should traps be used to monitor WCR beetles in soybean? Each week for 6 weeks, or until the beetle threshold is reached, remove the traps, and place new ones just above the soybean canopy. Count and record the number of rootworm beetles on each trap. To determine the average number of beetles/trap/day, add the numbers for the 6 traps in each field, divide that number by 6, and then divide by the number of days the traps have been in the field. Although a 7-day sampling period is preferred, be sure to divide by the actual number of days the traps were in the field to determine the average. When do trap counts indicate the need for a management tactic? Where can I get the traps? Where can I get more WCR information?
Borers in Stems of Large Weeds– (John Obermeyer) Phone calls and questions at field days make it obvious that many pest managers are finding borers in the stems of giant and common ragweeds, marestail, and lambsquarters. The extensive tunneling in these weeds prevented sufficient translocation of the herbicide to kill the plants. The larvae most likely in the giant ragweed is the stalk borer, this was already discussed in Pest&Crop #15, June 28, 2002. Another worm, which looks much like European corn borer, has not been positively identified. Knowing that European corn borer has over a hundred hosts, it is indeed possible. Another borer, the smartweed borer, is a close cousin and may be the culprit in some cases. No matter the species of insect, the results are the same … poor weed control.
|
|||||||||
Soybeans and Growth Regulators?– (Glenn Nice and Thomas Bauman)
As the summer moves along the Purdue’s Plant & Pest Diagnostic Laboratory starts to get a lot of soybeans that have puckered and strapped leaves. This was also the case last year. In many cases growth regulator injury is suspected. However, there can be more to the story. Growth Regulators Leaf cupping and strapping (see figures 1 and 2) can result from growth regulator (Banvel, Clarity, Distinct, 2,4-D) injury. There are a couple of ways in which a soybean crop can come in contact with a growth regulator herbicide. One way is in the form of drift. Drift may take the form of particle drift, the movement of droplets, or vapor drift, the movement of volatized herbicide. This type of injury often appears this time of year around the time corn herbicides such as Clarity, Banvel, Marksman, Stinger, and 2,4-D are being applied POST. Corn and soybean fields are often next to each other or across the road from each other. This provides, depending on conditions (wind direction, relative humidity, wind speed, application pressure, etc), a good potential for injury from drift. Another way that growth regulator herbicides can come in contact with soybean is in the form of tank contamination. Residues left in the tank can be brought into solution naturally or from solvents found in some of the herbicides. The Banvel label gives good instructions for cleaning tanks and application equipment after the use of growth regulator herbicides. To prevent this many custom applicators use separate tanks, one for corn herbicides and one for soybean. However, the reason for the large amount of soybean leaf abnormalities is not clear cut. Some soybean fields do not fit the drift scenario (See “Is it Drift or Not?” below). In these cases, the causes of the foliar abnormalities are not well understood. This is partially due to difficulty in doing research on this phenomenon. Secondly, correlations between environmental conditions or agricultural practices don’t always seem to be consistent. This has lead to a few possibilities but not much data. POST Applications One possibility is that components of a POST application might be doing it. In 2000, Aaron Hager and Christy Spraque (University of Illinois) pointed out that many of the samples sent to them had been treated with a herbicide that translocated, but stated that this was not always the case. An additive is usually used with POST applications and may have some influence. Purdue’s soybean extension specialist Elsworth Christmas has suspected on occasion that the use of AMS may have had some part in these soybean abnormalities. Environment Like many aspects of agriculture the plant growth environment probably plays a part. The possibility that has been proposed is that this could be a physiological response to environment. That poor growing conditions or sudden fluctuations in temperature can cause problems with hormone regulation leading to symptoms observed. The search for connections with hot temperatures, hot and humid conditions, cold wet followed by hot dry conditions, or a multitude of environmental patterns and cues is occurring. However, there is no data at this time that I am aware of confirming this. Other Possibilities Questions have come up concerning certain insects such as spider mites or leafhoppers. Also micronutrients in the form of manganese toxicity or boron toxicity have been written about. More complicated than that is that it may be a combination of things, which would give some support to the difficulty on doing research on this condition. Drift or Not? Indiana is not new to problems experienced with drift of herbicides. Many of the cases seen may be a result of drift. Standing in a field looking at cupped soybean, the question is, ‘is it drift or not’? I use the three strike method. 1)Are the symptoms suggestive of growth regulator herbicides? The cupping and strapping (Fig 1 and 2) are symptoms that you would see from a 2,4-D (many) or dicamba (Banvel, Clarity, or Distinct). 2) Look for patterns in the field. Generally injury from drift will have a pattern. Particle drift will induce more damage closer to the source of the drift. It will cross over rows and reduce the farther away from the source. The pattern may become less clear if there are several sources of drift. Records of wind directions from local weather service will aid in this aspect. If the injury borders are defined by rows then suspicion is placed on mechanical or tank contamination. Vapor drift is often more difficult to find a pattern. This is the movement of the molecules as a gas and can travel farther and after the application. One thing to note is often injury will be worse in the low portions of the field. However, this is not always evident and a third criterion must be examined. 3) What are the weeds doing? Look at other plants in the area, specifically in the ditch or areas where an application from either field would require drift to reach it. A strong indication of drift would be if weeds (velvetleaf, ragweed, mustard, pigweed) between the suspected source of drift and the soybean injury show symptoms such as stem or petiole twisting, leaf cupping or strapping. Look in areas where applications from the fields themselves would have less of an effect; areas where only drift could reach the injured weeds. One strike leads me to question weather it is drift or not. I have seen fields that showed cupping and strapping of the leaves in an even pattern across the field, but none of the weeds showed any such injury. This leads me to suspect something else is at work. Will This Effect Yield? Many times drift may not reduce yield as crops will recover. In other situations yield is reduced. In many cases, symptoms appear on older leaves and the new growth appears normal. If the symptoms appear due to a growth regulator, foliar symptoms are reported to occur at rates that are generally lower than yield reducing rates. However, the chance of yield loss increases if the soybean is affected during late flowering. If this happens to you, keep a close eye on yield in smaller areas to do comparisons may shed some light on the effects on yield. In any drift situation remember the four ‘R’s. Recognize, Respond, Resolve, and remember. Recognize the potential for possible drift before spraying. The best way to deal with a drift situation is not to let it happen. If a neighbor contacts you with a drift complaint, respond immediately and courteously. Resolve the problem in a peaceful manner. No one wants an ugly court battle. Remember to think and look before you spray. When complaining about a drift incident, remember that you could be on the other end of it next year. Always remember to read and follow pesticide labels.
|
|||||||||
Brown Spot of Corn– (Gregory Shaner)
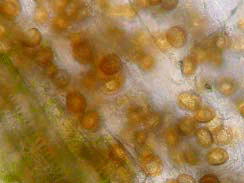
Gail Ruhl and I diagnosed brown spot on a corn sample from southern Indiana submitted to the Plant and Pest Diagnostic Lab last week. Leaves of these plants had small, golden-brown spots. Over most of the affected leaf tissue, the spots had coalesced to form large patches of dead tissue. Symptoms were most severe on the apical half of the leaf. The patches of dead tissue tended to occur in bands across the leaf with more or less healthy tissue in between. We also observed strings of black circular lesions near the midrib. It was in this tissue that Gail was able to find sporangia of the brown spot fungus, Physoderma maydis (see figures below). Although the disease is reported to occur in the Midwest, we have rarely seen it in the PPDL. Brown spot is more common in tropical areas, where there are long periods of moisture and high temperatures. Reduced tillage also favors the disease, because the sporangia produced by the fungus in infected plants survive in crop residue. Sporangia of Physoderma maydis embedded in tissue near the midrib. Photos by Gail Ruhl, Purdue Plant and Pest Diagnostic Laboratory
Soybean Rust– (Gregory Shaner)
I have received questions about soybean rust and the threat it might pose to Indiana’s soybean crop. This disease has never been found in the continental U.S. It has been known for many years in Asia, where soybeans originated. Recently its range has expanded, and it is now in South America, as well as several counties in Africa. Rust diseases are certainly not strangers to the U.S. Rust fungi have attacked many of our crops for years, including wheat, oats, barley, and corn. There are also rust diseases of vegetables, fruit crops, and ornamentals. However, these rust fungi are not related to the rust fungus that attacks soybeans. The rust fungi are a highly specialized group. There are about 7000 known species. Each one infects only a few related host plant species. For example, the leaf rust fungus that infects wheat can infect a few other grasses, but does not infect oats, barley, or other crops. The rust fungi that infect corn do not infect small grains. The rust fungus that infects soybeans can infect a number of other species in the bean family (Fabaceae), but most of these other hosts are only found in tropical regions. Unlike many leaf-infecting fungi that hibernate in crop residue and then produce new spores on that residue the following spring, rust fungi require a living host plant to produce the microscopic spores that initiate infection. The rust fungi that infect various cereal crops in Indiana hibernate in southern states. During the spring, rust spores produced on plants in the South are carried north and infect the crops here. Rust spores are well adapted for long-distance dispersal. There are well-documented examples of rust spores moving across oceans. This is probably how the soybean rust fungus made the trip from Asia to South America. The soybean rust fungus does not infect seed, so it is doubtful that it could move between South America and the U.S. on seed. Although the fungus cannot grow or produce spores on dead soybean leaf tissue, spores that form in living plant tissue may remain viable for several weeks, depending on environmental conditions. Thus, bits of leaf tissue that might accompany seed could harbor viable spores of the fungus. Another means of long distance transport could be living plants of ornamental host species that are shipped between these two regions. The Animal and Plant Health Inspection Service (APHIS) within the U.S. Department of Agriculture has published a Web site that contains useful information about soybean rust, including several images of infected plants and the fungus. It can be found at: <http://www.aphis.usda.gov/ppq/ep/pestdetection/soybean_rust/detection3.html>. Remember, soybean rust has not yet been found in the continental US, so it is unlikely that the disease is in Indiana. However, if you see soybeans with the symptoms shown in the photos on the USDA website, you may wish to send a sample to the Purdue University Plant and Pest Diagnostic Laboratory (PPDL), 1155 LSPS, Purdue University, West Lafayette, IN 47907-1155 for diagnosis. Leaf samples should be pressed between pieces of dry paper towel or cardboard, and mailed using overnight or 2-day service to avoid sample deterioration during transit. Contact your local county Cooperative Extension office or call the PPDL at 765-494-7071 for a copy of the PPDL submission form. There is a fee charged for samples submitted to the PPDL.
|
|||||||||
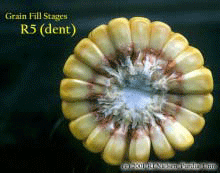
Grain Fill Stages in Corn– (Bob Nielsen) The grain fill period begins with successful pollination and initiation of kernel development, and ends approximately 60 days later when the kernels are physiologically mature. During grain fill, the developing kernels will be the primary sink for concurrent photosynthate produced by the corn plant. What this means is that the photosynthate demands of the developing kernels will take precedence over that of much of the rest of the plant. In essence, the plant will do all it can to ‘pump’ dry matter into the kernels, sometimes at the expense of the health and maintenance of other plant parts including the roots and lower stalk. A stress-free grain fill period can maximize the yield potential of a crop, while severe stress during grain fill can cause kernel abortion and lightweight grain. Kernel development proceeds through several relatively distinct stages.
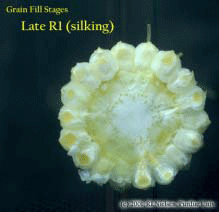
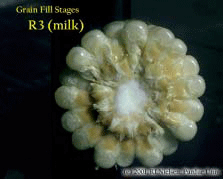
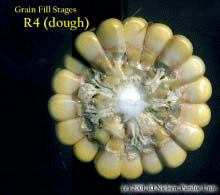
Silking Stage (Growth Stage R1). Some may argue whether silking should be labeled as a kernel growth stage, but nonetheless silk emergence is technically the first identifiable stage of the reproductive period. Silks remain receptive to pollen grain germination up to 10 days after silk emergence. Silk receptivity decreases rapidly after 10 days if pollination has not yet occurred. Natural senescence of silk tissue over time results in collapsed tissue that restricts continued growth of the pollen tube. Silk emergence usually occurs in close synchrony with pollen shed, so that duration of silk receptivity is normally not a concern. Failure of silks to emerge in the first place (for example, in response to silkballing or severe drought stress) does not bode well for successful pollination. Kernel Blister Stage (Growth Stage R2). About 10 to 14 days after silking, the developing kernels are whitish ‘blisters’ on the cob and contain abundant clear fluid. The ear silks are mostly brown and drying rapidly. Some starch is beginning to accumulate in the endosperm. The radicle root, coleoptile, and first embryonic leaf have formed in the embryo by the blister stage. Severe stress can easily abort kernels at pre-blister and blister stages. Kernel moisture content is approximately 85 percent. Kernel Milk Stage (R3). About 18 to 22 days after silking, the kernels are mostly yellow and contain ‘milky’ white fluid. The milk stage of development is the infamous ‘roasting ear’ stage, that stage where you will find die-hard corn aficionados standing out in their field nibbling on these delectable morsels. Starch continues to accumulate in the endosperm. Endosperm cell division is nearly complete and continued growth is mostly due to cell expansion and starch accumulation. Severe stress can still abort kernels, although not as easily as at the blister stage. Kernel moisture content is approximately 80 percent. Kernel Dough Stage (R4). About 24 to 28 days after silking, the kernel’s milky inner fluid is changing to a ‘doughy’ consistency as starch accumulation continues in the endosperm. The shelled cob is now light red or pink. By dough stage, four embryonic leaves have formed and about 1/2 of the mature kernel dry weight is now in place. Kernel abortion is much less likely once kernels have reached early dough stage, but severe stress can continue to affect eventual yield by reducing kernel weight. Kernel moisture content is approximately 70 percent. Kernel Dent Stage (R5). About 35 to 42 days after silking, all or nearly all of the kernels are denting near their crowns. The fifth (and last) embryonic leaf and lateral seminal roots form just prior to the dent stage. A distinct horizontal line appears near the dent end of the kernel and slowly progresses to the tip end of the kernel over the next 3 weeks or so. This line is called the ‘milk line’ and marks the boundary between the liquid (milky) and solid (starchy) areas of the maturing kernels. Severe stress can continue to limit kernel dry weight accumulation. Kernel moisture content at the beginning of the dent stage is approximately 55 percent. Physiological Maturity (R6). About 55 to 65 days after silking, kernel dry weight usually reaches its maximum and kernels are said to be physiologically mature and safe from frost. Physiological maturity occurs shortly after the kernel milk line disappears and just before the kernel black layer forms at the tip of the kernels. Severe stress after physiological maturity has little effect on grain yield, unless the integrity of the stalk or ear is compromised (e.g., ECB damage or stalk rots). Kernel moisture content at physiological maturity averages 30 percent, but can vary from 25 to 40 percent grain moisture. Harvest Maturity. While not strictly a stage of grain development, harvest maturity is often defined as that grain moisture content where harvest can occur with minimal kernel damage and mechanical harvest loss. Harvest maturity is usually considered to be near 25 percent grain moisture. Related Reference: Ritchie, S.W., J.J. Hanway, and G.O. Benson. 1993. How a Corn Plant Develops. Iowa State Univ. Sp. Rpt. No. 48. On the Web at <http://maize.agron.iastate.edu/corngrows.html> (last verified 7/17/02).
When and How Can I Estimate Corn Yields?– (Bob Nielsen) Corn growers are especially keen on estimating their grain yield potential this year for two reasons. The first reason is the recent rally in grain prices and the uncertainty about whether the rally will continue in coming weeks or fade down the stretch. The other reason lies with corn growers’ uncertainty about the effects of this year’s multitude of crop stresses on the eventual grain yield. Obviously, to satisfy the desire to sell grain at higher prices requires that there be grain to sell. Therein lies the interest in field-by-field yield estimations to help develop their grain marketing plans. A number of yield prediction methods exist, but the one most commonly used in the field is probably the Yield Component Method. Sometimes referred to as the Slide Rule Yield Calculator, this method was developed by the University of Illinois many years ago. The Yield Component Method can be used well ahead of harvest; at least by the time that kernel development has reached the late milk to early dough stages (R3 to R4). Under “normal” conditions, this point in kernel development occurs about 25 days after pollination is complete (see my related article on grain fill stages). Estimates made earlier in the kernel development period risk being overly optimistic if subsequent severe stresses cause unforeseen kernel abortion prior to about the roasting ear stage (R3 or milk). Crop uniformity greatly influences the accuracy of any yield estimation technique. The less uniform the field, the greater the number of samples that should be taken to estimate yield for the field. There is a fine line between fairly sampling disparate areas of the field and sampling randomly within a field so as not to unfairly bias the yield estimates up or down. 1. At each estimation site, measure off a length of row equal to 1/1000th acre. For 30-inch rows, this equal 17 ft. 5 in. For 36-inch rows, this would equal 14 ft. 6 in. For other row spacings, divide 43560 by the row spacing (in feet) and then divide that result by 1000. For example, let’s say you counted 24 harvestable ears at the first sampling site. Sampling every 5th ear resulted in an average row number of 16 and an average number of kernels per row of 30. The estimated yield for that site would (24 x 16 x 30) divided by 90, which equals 128 bu./ac. Repeat the procedure in as many sites within a field as you deem representative. Calculate the average yield for all the sites to estimate the yield for the field. Remember that this method for estimating pre-harvest grain yield in corn indeed provides only an estimate. Since kernel size and weight will vary depending on hybrid and environment, this yield estimator should only be used to determine “ballpark” grain yields. Yield will be overestimated in a year with poor grain fill conditions (e.g., low kernel size and weight from a drought year) and underestimated in a year with excellent grain fill conditions (e.g., larger kernel size and weight from non-stress grain fill periods). For example, if you believe that kernel weight will be less due to stress during grain fill, you may elect to replace the value of ‘90’ in the equation with ‘100’ to reflect the potential for smaller and lighter kernels (i.e., more kernels per bushel). Conversely, in a good crop year, you may elect to replace the value of ‘90’ in the equation with ‘80’ to reflect the potential for larger and heavier kernels (i.e., fewer kernels per bushel). The Yield Component Method for estimating corn grain yield is probably accurate within plus or minus 30 bushels of the actual yield. Obviously, the more sampling performed within a field, the more accurately you will ‘capture’ the variability of yield throughout the field. Use the yield estimates obtained by this method for general planning purposes only. Related Reference: Nielsen, RL (Bob). 2002. Grain Fill Stages in Corn [Online]. Purdue University, Agronomy Dept., W. Lafayette, IN 47907-1150. On the Web at <http://www.kingcorn.org/news/articles.02/Grain_Fill-0723.html > (Verified 7/23/02). Don’t forget, this and other timely information about corn can be viewed at the Chat ‘n Chew Café on the World Wide Web at <http://www.kingcorn.org/cafe>. For other information about corn, take a look at the Corn Growers’ Guidebook on the World Wide Web at <http://www.kingcorn.org/ >.
|
|||||||||
|
|||||||||


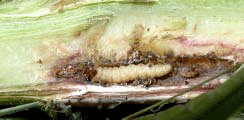


 Dark lesions near midrib that contained sporangia of the fungus.
Dark lesions near midrib that contained sporangia of the fungus.