Pest & Crop Newsletter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
2002 Insect Pests, Expect the Unexpected- (John Obermeyer, Rich Edwards, and Larry Bledsoe)
There is no “typical” for this growing season, including insect pests of field crops. With planting and crop development completely askew this year, bizarre occurrences should be expected. Calls this week didn’t disappoint. Bryan Overstreet, Jasper County Co-op, called about millipedes feeding on seedling corn and Dwain Rule, Midland Co-op, reported spotted cucumber beetles defoliating 2-3 leaf corn. This is just the beginning of what may be an interesting year for pest managers, load the camera up with film! Non-crop insects are often forced to feed on emerging crops where very weedy fields existed. These miss-placed insects were feeding on weeds, until their host was taken away by tillage or herbicides. This will often catch the attention of those monitoring fields and provide some excitement. However, these occasional invasions are often short-lived as the insects starve to death or migrate to greener “pastures.” Normally this time of the year, Ron Blackwell, IPM Survey Specialist, is surveying for first generation corn borer damage in southern Indiana. We know from black light traps that corn borer moths have been flying which is right on cue. But, with corn either just being planted or ankle high, the moths are obviously passing over our yellow corn for one of many alternative hosts for egg laying. Instead, Ron is sampling emerging cornfields for black cutworm and finding none. This certainly has been a year to test the theory about increasing concerns with white grubs over the past several years. Though we have speculated about many possibilities, earlier planting trends has been our chief suspect. So far, with only 13% of the planted corn emerged (Indiana Ag Statistics Service), phone calls about these pests have been nil. The remaining corn to emerge, or to be planted, should develop quickly and leave little time for these critters to nip the seedlings to death. The grubs are there, but seem content feeding on dead and/or decaying matter. This reduced interface between these soil pests and seedlings should result in fewer damaged plants. Appears to be rough year for rootworms. Though we still have not documented egg hatch in west central Indiana (our indicator corn couldn’t get planted) we suspect that it has started in southern Indiana. Hatching larvae are likely to find either saturated soils or no corn roots at all. This obviously leads to drowning or starvation of larvae, which means fewer beetles this summer. This should be an excellent year for producers, consultants, agronomists, etc. to implement monitoring programs for western corn rootworm beetles in soybean fields. More on trapping with yellow sticky traps later. Pest managers in 2002 should be exceedingly alert and open to many possibilities as they monitor fields. Don’t throw away the pest management textbooks, but recognize the fact that insects can’t read. Happy scouting!
Corn Flea Beetle, Thick in Some Areas– (John Obermeyer, Rich Edwards, and Larry Bledsoe)
There is no doubt that the mild winter has contributed to the numbers of corn flea beetles being observed in some areas of Indiana. This tiny (1/16”), shiny black beetle feeds on corn leaves by stripping off the top layer of plant tissue. This feeding leaves gray to brown lines or “tracks” etched on the leaf surface. Heavily infested plants may appear gray as their leaves shrivel and die.
On seedling dent corn, control may be necessary if 50% of the plants inspected show severe corn flea beetle feeding damage (plants begin to look silvery or whitish, or leaves begin to die), approximately 5 or more corn flea beetle per plant are found, and poor growing conditions are causing slow corn growth (e.g., cool temperatures, herbicide damage). Normally, once a corn plant has several leaves and growing conditions improve it is no longer susceptible to significant corn flea beetle damage. Corn flea beetle may also transmit the bacterium that causes Stewart’s wilt as it feeds. This can be a serious problem, especially on sweetcorn and seed corn inbreds. In sweetcorn, the disease may result in ears that are smaller than normal, or some infected plants may die. In seed production fields, severe leaf blight may cause lightweight chaffy ears, plus increase the likelihood of stalk rots. The beetles alone are seldom severe enough to kill plants although in combination with the disease, such as noted above for sweetcorn, they may. In seed production fields where highly susceptible inbreds are utilized, treatment is probably justified if corn flea beetles are noted.
Potato Leafhoppers... They're Here– (John Obermeyer)
By monitoring my windows last night (yes, bug guys do that) it became obvious that potato leafhoppers have arrived. Although these leafhoppers are of no significance at this time, it does indicate the beginning of the 2002 population. As the season progresses, populations will increase and may reach economic levels after the first and subsequent cuttings. As if alfalfa weevil and miserable haying conditions weren’t enough for the alfalfa crop this year already! Stay tuned.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Head Scab in Wheat– (Gregory Shaner)
Head scab of wheat is a wet weather disease, so it’s not surprising that it is showing up. I saw some scab in a cultivar trial at the Southwest Purdue Ag Center a week ago. Heads were entirely or partially bleached on affected plants. Most cultivars were in the watery ripe stage of development. Scab was not severe. In the cultivar with the greatest incidence of scab, only 5 out of 10,000 heads showed symptoms. That’s the good news. The bad news is, although flowering is the most vulnerable period for infection, infection can occur anytime during early grain development, perhaps as late as early dough. So, the incidence of scab may increase where rains have continued to fall after wheat completed flowering. We had a major scab epidemic in 1996. In the cultivar Caldwell, grown at Lafayette, 66% of the heads were blighted by about 3 weeks after flowering. At that location, rain fell on 6 consecutive days, beginning with the initiation of flowering. Most wheat in Indiana has not been subjected to that much continuous rainfall during flowering this year, but some rain has fallen during flowering, and it continues to fall. Temperature also influences scab development. A study done many years ago suggested that the optimum temperature for disease development was 77°F, and that at 59°F no infection would occur. However, during those rainy days in Lafayette in 1996, high temperatures were only in the low 60s and low temperatures were in the low 50s, yet severe disease developed. This is not unlike the temperatures that were common when wheat in southern and central Indiana began flowering. I am not predicting that we will have as much scab as we did in 1996, but it is possible that the disease will be evident in many wheat fields. The time to start checking for scab is when wheat is just getting into the milk stage of development (Feekes stage 11.1). Often, only a portion of an infected head will be blighted initially. This corresponds to the spikelets that were flowering when infection occurred. More of the head will become blighted with time. The fungus grows into the rachis and invades other spikelets. On a susceptible cultivar, the entire head will become blighted. Grain, especially in the florets that were infected early, will be severely shriveled. The foliage on scabby heads will not necessarily be diseased. The upper two leaves and the peduncle (neck) may be a healthy green. The fungi that cause scab (Fusarium graminearum and some other species of Fusarium) are not foliar pathogens. If leaf disease is present, it is more likely to be leaf blotch, caused by Septoria tritici or Stagonospora nodorum. These fungi are not involved in head scab.
Stripe Rust in Wheat– (Gregory Shaner)
I have found stripe rust in wheat both at the Southwest Purdue Ag Center in Knox County and at the Purdue Agronomy Research Center in Tippecanoe County. It’s very likely that the rust can be found in many other areas as well. As the disease name suggests, pustules of this rust form stripes on the leaf. The stripes are bounded by major veins in the leaf, so are a little under 1/8 in. wide, but can extend for an inch or more in length. Within these stripes, there are numerous pustules. Each pustule is a mass of fungal spores that have burst through the leaf epidermis. The pustules are yellow. In Europe, the disease is known as yellow rust because of this distinctive color. The stripes and pustule color serve to distinguish this rust from leaf rust (orange pustules) and stem rust (brick red pustules). Of these three rusts on wheat, stripe rust is favored by the coolest weather. Normally, weather in Indiana passes from winter to summer too quickly for stripe rust to thrive. The cool spring this year has evidently favored stripe rust. In the U.S., stripe rust is mainly a problem in the Pacific Northwest, California, and some of the Rocky Mountain States. In recent years, the disease has occurred frequently in the southeast U.S. Many wheat fields in Arkansas were severely damaged by stripe rust last year. In the Pacific Northwest, initial infection often occurs in the fall. Rust will develop in local areas in a field, around points where initial infection occurred. As the season progresses, the rust spreads to upper leaves and to other plants. The stripe rust I have seen this spring in Indiana is on flag leaves, which indicates that the infections occurred from airborne spores blown in recently from southern states. So far, the rust is focal. That is, a few adjacent plants will have fairly severe rust, but most of plants in a field do not show symptoms. The cool weather that prevailed until a few days ago suggests that these infection foci may enlarge over the next couple of weeks. It is difficult to predict if the rust will become severe enough to cause damage. Because stripe rust is not a disease we normally see, our varieties are probably all more or less susceptible (wheat breeders, understandably, do not to breed for resistance to diseases that do not occur in their area). If anyone encounters a severe case of stripe rust in a field of wheat, I would like to hear about it. You can reach me by e-mail at <shanerg@purdue.edu>.
Switching From Corn to Soybeans- The Disease Angle– (Gregory Shaner)
There has been a lot of discussion recently about the wisdom of switching from corn to soybeans as the delay in corn planting continues. Various specialists at Purdue have presented the pros and cons of doing this. Both the pro and con articles have referred to soybean diseases as an important consideration in changing planting plans. If many acres are switched, this will mean planting soybeans in fields that were planted to soybeans last year. I want to elaborate on what can happen with diseases if soybeans follow soybeans. The major diseases of soybeans in Indiana and surrounding states are caused by soilborne pathogens. These pathogenic microorganisms survive in soil, often through production of special survival structures. These microorganisms, in no particular order, include Heterodera glycines (soybean cyst nematode), Phytophthora sojae (Phytophthora root rot), Fusarium solani f. sp. glycines (sudden death syndrome), Sclerotinia sclerotiorum (white mold), and Phialophora gregata (brown stem rot). The soybean cyst nematode produces cysts that can survive several years in soil. Phytophthora sojae produces oospores and Sclerotinia sclerotiorum produces sclerotia. These can also survive for many years in the absence of soybeans. Fusarium solani f. sp. glycines and Phialophora gregata survive as mycelium in soybean residue or produce a spore that can survive in soil. When these organisms are in a field and a susceptible cultivar of soybean is grown, they multiply (from a pathogen’s point of view, the disease it causes is secondary; it is simply using the host plant as a food source to support its growth and multiplication). When a nonhost such as corn is grown in the field the following year, these pathogens cannot multiply, and may die out to some extent. The general increase in problems with all of these diseases in recent years is thought to be a result of the shift to a soybean-corn rotation in place of rotations in which soybeans were grown only at longer intervals in a field. So, what does it mean if a grower goes back into a field with soybeans in 2002 that was planted to soybeans in 2001? If any of the pathogens mentioned above was not in this field, a second year of soybeans does not mean it will suddenly appear. However, all of these pathogens are widely distributed in Indiana. A danger from second-year soybeans is that in a field with only a small or moderate pathogen population, and hence a history of only mild or moderate disease problems, a second year of soybeans could lead to a great increase in pathogen populations. A minor problem with disease could become a major problem this year. Once these high pathogen populations have developed, it can take a long time to bring them back down, particularly by going back to a rotation in which soybeans are grown every other year. An even greater danger is to plant second-year soybeans in a field that already has a history of serious disease problems. None of these pathogens is capable of long-distance dispersal under its own power. The diseases they cause typically appear in some areas of a field and not others. Anything that moves soil can disperse these pathogens, however. Farming operations are some of the most efficient agents of dispersal. Tillage equipment, planters, tractor tires – anything that moves soil or plant material – can increase the area in a field that is affected by a soilborne disease. A second year of soybeans can result in a greater proportion of a field being infested with a pathogen. History of disease in a field, choice of soybean cultivar, crop management practices, and weather all play a role in determining whether disease will be a problem. But, the risk of a disease problem can only be greater with second-year soybeans.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
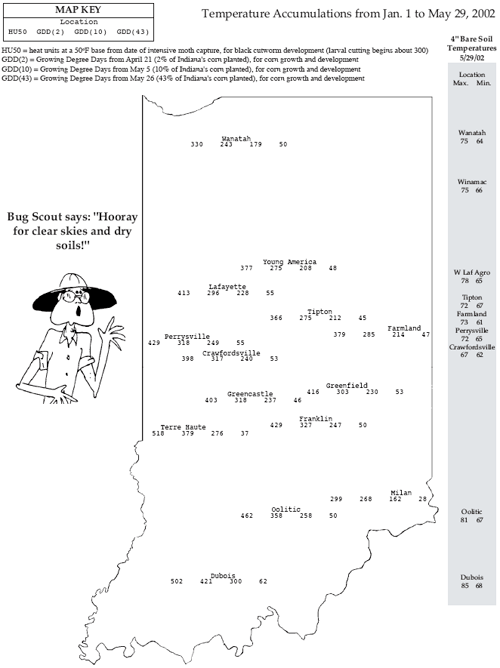
What Exactly Do You Mean by "Growing Degree Day"?– (Bob Nielsen)
Growth and development of corn are strongly dependent on temperature. Corn develops faster when temperatures are warmer and more slowly when temperatures are cooler. For example, a string of warmer than normal days in late spring will encourage faster leaf development than normal. Another example is that a cooler than normal grain filling period will delay the calendar date of grain maturity. The phrases “string of warmer than normal days” and “cooler than normal grain filling period “ can be converted mathematically into measures of thermal time by calculating the daily accumulations of heat. Commonly used terms for thermal time are Growing Degree Days (GDDs) or heat units (HUs). Different methods exist for calculating heat units depending on a) the crop or biological organism of interest and b) the whim or personal preference of the researcher. In Purdue’s Pest&Crop Newsletter (<http://www.entm.purdue.edu/Entomology/ext/targets/newslett.htm>), you will often see more than one type of calculated GDD or HU reported depending on the insect or crop of interest. The calculation method most commonly used in the U.S. for determining heat unit accumulation relative to corn phenology is the formula first suggested by the National Oceanic and Atmospheric Administration in 1969 and labeled as the ‘Modified Growing Degree Day’ formula in 1971. This method calculates daily accumulation of GDDs as the average daily temperature minus 50°F. The ‘modification’ refers to the limits imposed on the daily maximum and minimum temperatures allowed in the calculation. Daily maximums greater than 86°F are set equal to 86 in the calculation of the daily average temperature. Similarly, daily minimums less than 50°F are set equal to 50 in the calculation. Example 1: If the daily maximum temperature was 80°F and the minimum was 55°F, the GDD accumulation for the day would be ((80 + 55)/2) – 50 or 17.5 GDDs. Example 2 (Illustrating the limit on daily maximums): If the daily maximum temperature was 90°F and the minimum was 72°F, the GDD accumulation for the day would be ((86 + 72)/2) – 50 or 29 GDDs.
What Exactly Do You Mean by "Leaf"?– (Bob Nielsen)
Growth staging in corn can be confusing; some even claim it’s a shell game. The confusion exists because there is no universally accepted method to stage corn. Agronomists have their method, hail insurance adjusters have theirs, and herbicide labels are in between the two.
Leaf Collar Method. Agronomists determine leaf stage in corn by counting the number of leaves on a plant that have visible leaf collars, including the first short rounded-tip leaf. The leaf collar is the light-colored ‘band’ visible at the base of an exposed leaf blade. ‘Droopy’ Leaf Method. Hail insurance adjusters also begin counting with the first short leaf, but continue counting beyond the uppermost visible collar to that leaf which is 40 to 50 percent exposed. In knee-high corn or older, the tip of this leaf is typically also pointing downward, hence the name ‘droopy’ leaf method. The few post-emergence corn herbicide labels that refer to corn leaf stage typically use the ‘droopy’ leaf method, except that the first short leaf is not counted. Leaf Collar vs. Droopy Leaf. Up to the 5- to 6-leaf collar stage, the leaf collar method will typically result in a leaf stage that is one less than the ‘droopy’ leaf method. After corn reaches 18 to 24 inches in height, the leaf collar staging method will typically result in a leaf stage that is two less than the ‘droopy’ leaf method. As corn plants develop, the lower few leaves typically die or are ripped from the stem by expansion of the stalk or by developing roots. Consequently, growth stage identification can be difficult on older plants when you aren’t sure which leaves to begin counting first. But, not to worry, growth staging can still be accomplished! First, dig or pull a plant without breaking the stalk. With a knife, split the stalk down the middle, completely through the root ball. Look for the first noticeable internode (the whitish area between the ‘woody’ horizontal stalk nodes) above the triangular ‘woody’ base of the stalk. This internode’s length is typically only 1/2 to 3/4 inch. Carefully determine which leaf’s sheath attaches to the node immediately above the first noticeable internode. This leaf is usually Leaf #5. Once Leaf #5 is identified, then stage the plant by counting the remainder of the leaves up the plant that have visible leaf collars. If you want to determine growth stage in order to use a defoliation/yield loss chart, then remember to add 2 to that number to equal the ‘droopy’ leaf method. The usefulness in understanding the differences between the leaf collar and ‘droopy’ leaf methods lies in the fact that the defoliation/yield loss chart used by hail insurance adjusters depends on the ‘droopy’ leaf method. That chart is reproduced in Purdue’s Corn & Soybean Field Guide, Extension publication # ID-179. Severe hail damage often limits your ability to spot true ‘droopy’ leaves, whereas leaf collars are often still identifiable. Thus, you can usually stage a damaged crop by the leaf collar method, then add one or two more leaves to the count in order to use the defoliation chart.
Predicting Leaf Stages in Corn– (Bob Nielsen)
Phenology: A branch of science dealing with the relations between climate and periodic biological phenomena (Webster’s New Collegiate Dictionary, 1973) Predicting when a field of corn will reach particular leaf stages can be useful for planning post-emergence applications of certain herbicides and sidedress N fertilizer, especially if your farming operation is so large that field inspections are difficult to work into your busy schedule. Recent research by one of my former graduate students helps fine-tune our ability to predict the portion of corn phenology that we call leaf stage development. Review of Concepts. For a refresher on how corn leaf staging is done, be sure to read the accompanying article “What Exactly Do You Mean by ‘Leaf'?." Another topic that probably needs reviewing is the concept of heat units (HUs) or growing degree days (GDDs). The concept is important because corn phenology is very dependent on temperature. Heat unit or GDD calculation for corn phenology is described in an accompanying article “What Exactly Do You Mean by ‘Growing Degree Day?'. Predicting Corn Phenology. Given an understanding of corn leaf stage development and heat unit calculation, you are now prepared to predict what leaf stage of development a particular field is at given its planting date and temperatures since planting. It is useful if you know when the crop emerged, but if not you can estimate that event also. Corn emergence typically requires 100 to 150 GDDs. Based on my former graduate student’s research (Kirby Wuethrich. 1997. Vegetative and Reproductive Phenology of Fourteen Hybrids of Dent Corn (Zea mays L.). Purdue Univ.), we now know that corn leaf developmental rates can accurately be described in two phases. From emergence to leaf stage V10 (ten visible leaf collars), leaf emergence occurs approximately every 85 GDDs. From leaf stage V10 to the final leaf, leaf emergence occurs more rapidly at approximately one leaf every 50 GDDs. Example 1: A field was planted on April 28 and emerged on May 5. Since May 5, approximately 535 GDDs have accumulated. Based on our research data, the estimated leaf stage for the crop (without looking at the field, mind you) would be between V6 (6 leaves x 85 GDDs = 510 GDDs) and V7 (7 leaves x 85 GDDs = 595 GDDs). Example 2: A field was planted on April 28, but you do not know exactly when it emerged. Since planting, approximately 785 GDDs have accumulated. If you assume that the crop emerged in about 125 GDDs, then the estimated leaf stage for the crop would be between V7 and V8. The estimation stems from first subtracting 125 from 785 to account for emergence, then dividing the result (660) by 85 to equal 7.8. Example 3: A field was planted on April 28 and emerged on May 5. Since May 5, approximately 1200 GDDs have accumulated. Your experience with these calculations tells you that the crop is likely beyond V10 (equal to 10 x 85 or 850 GDDs). First subtract 850 from 1200 (knowing the crop is at least at V10). Divide the result (350) by 50 to equal 7 additional leaves for a total estimated leaf stage of V17. Keep in mind that estimates of leaf stage development are only that, estimates. One of the factors that most influences the accuracy of these estimates is the existence or not of other growth-limiting stresses. However, what I have described here will put you in the proverbial ballpark in determining which fields are at which leaf stages. Don’t forget, this and other timely information about corn can be viewed at the Chat ‘n Chew Café on the World Wide Web at <http://www.kingcorn.org/cafe>. For other information about corn, take a look at the Corn Growers’ Guidebook on the World Wide Web at
Prognosis Poor for Corn "Leafing Out" Underground– (Peter Thomison, Ohio State University) Early growing season weather and seedbed conditions may create emergence problems resulting in corn seedlings leafing out underground. Seedings, which exhibit this abnormal emergence, may have a twisted appearance because internal leaves start expanding before the seeding has elongated. Several factors (or combination of factors) may be responsible for this abnormal growth. These factors may be characterized as environmental, chemical, or mechanical. Environmental conditions associated with underground leafing include light penetration, cold soils, or heavy rains soon after planting. When plants unfurl below the soil surface, they usually turn yellow and die. In a cloddy field where soil coverage of seed is poor and irregular, sunlight can reach the germinating seedling and induce leaf emergence beneath the soil surface. Also, heavy rains after planting can cause a hard crust, which makes emergence of small seedlings very difficult. As a result, bending and twisting of the seedling below the crusted layer often occurs. Planting the seed too deep, which may cause poor germination and emergence, may also result in premature unfurling of the corn. Certain herbicides such as Lasso and Dual, and the various premixes that contain their active ingredients can show similar symptoms (i.e., twisting, abnormal growth) when excessive rates are applied pre-emergence. Besides excessive rates, improperly closed seed furrows can allow the pre-emergence herbicide to come in direct contact with the seed. Excessive soil insecticide dosage or in-furrow placement of insecticide phytotoxic to seed can also cause twisting sprouts and abnormal leaf expansion underground. In addition, anhydrous and aqua ammonia N fertilizer injury has been associated with these symptoms. Certain corn hybrids are also more prone to this problem during emergence when unfavorable environmental conditions occur. Corn seedlings that exhibit abnormal unfurling symptoms during emergence will be unable to penetrate any but the loosest soil even if the crust is broken mechanically or softened by rain. Prompt treatment with a rotary hoe, weeder, spike tooth harrow or cultipacker may help break the crust and improve emergence. However, even when used carefully, these salvage operations can cause some damage to seedings, which are emerging normally. To minimize poor seedling emergence due to unfurling below the soil surface, watch for cloddy seedbeds, open seed furrows, and crusting surface soils after rains. Also check planting depth periodically and adjust accordingly during the planting operation and monitor herbicide and soil insecticide rates. (Published 5/28/02 in OSU’s C.O.R.N.)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
2002 DATES/LOCATIONS
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||