Pest & Crop Newsletter
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Western Corn Rootworm Winter Survival– (John Obermeyer, Rich Edwards, and Larry Bledsoe)
Three facets of corn rootworm biology will dictate the potential for rootworm damage to corn in 2001: 1) beetle egg laying in 2000, 2) the success of egg overwintering, and 3) larval survival and establishment. 1) Beetle egg laying in 2000: As already outlined in Pest&Crop #1, February 26, many first-year corn fields in northern Indiana will be at risk from rootworm damage because of last year’s western corn rootworm beetle numbers observed in soybean. This is also true of most corn following corn throughout the state. Simply put, more beetles last year laid eggs which means greater risk for root damage this year. 2) Egg overwintering and survival: We have received many questions this winter concerning the effects of temperature and tillage on overwintering rootworm eggs. After all, the Midwest has experienced some record low temperatures this winter. Rootworm egg survivability has been researched many times and in different ways in the past. To seek answers one must consider numerous variables such as; soil temperature, duration of temperature, soil moisture, soil type & texture, soil compaction, tillage type, residue cover, snow cover, and depth of egg. Researchers have conducted experiments in order to predict overwintering survival. A recent study was summed up as follows: “... factors such as soil moisture and temperature, which vary with soil depth and texture, and vary between and within years, make development of simple and highly predictable models of overwinter survival of western corn rootworm eggs difficult.” Lab experiments with controlled conditions have studied the effect of temperatures on hatch. A two year study found that temperatures of 23°F and 32°F for 8 weeks resulted in 80% and 17% mortality respectively. Soil temperature data from Purdue’s Agronomy Research Center, W. Lafayette during November and December (coldest 8 week period) averaged 35°F (minimums) at 4 inch (bare soil) depth. According to the above mentioned research, we shouldn’t rely on the winter’s cold to significantly decrease rootworm numbers. Tillage experiments (1940’s to present) to control rootworms have yielded a mixed bag of results. The strategy is to move the eggs near the soil surface where they will be exposed to lower soil moisture and temperature; desiccation causes high egg mortality. Knowing the depth of the overwintering egg is critical before tillage is considered as a control method. One study found 60% of the western corn rootworm eggs at a depth of 8-12 inches. Perhaps that is why researchers in one study concluded that “neither fall nor spring plowing can be recommended as a reliable method of controlling corn rootworms.” 3) Larval survival and establishment: Here too many factors influence the survival of the newly emerged larva. In short, barring environmental extremes such as drought or floods, hatched larva will find and begin feeding on corn roots within a few inches
Southwestern Corn Borer Spring Survey– (Ric Bessin, Doug Johnson, Wayne Mattingly, and Mike Smith, University of Kentucky) A survey of Southwestern corn borer damage and larval survival was conducted in Caldwell, Daviess and Henderson counties on March 6 and 7. These counties were selected because of the past history of damage in those areas. The purpose was to estimate the extent of SWCB damage as evidenced by basal stalk girdling. In addition, we wanted to estimate the survival of the larvae in the crowns of these damaged plants. In each county, four non-Bt corn fields were evaluated. Within each field, 10 groups of 10 plants were examined for girdling damage and presence of live SWCB larvae. An additional 50 damaged plants were examined for the presence of live SWCB larvae. This is the third year that we have conducted such a survey. Unlike the previous two winters, we had a severe cold period in December and January. Survival this winter is less than we estimated last year. Last year we estimated SWCB survival to be between 20 and 24% in those counties. In Daviess and Henderson counties the rate of survival is about half of what we saw the last two years. In Caldwell county it is considerably less. The average survival rate for Caldwell, Henderson and Daviess counties is 5.8%, 12.2%, and 9.2%, respectively. Why the difference among counties, it may be due to snow cover during the cold period. There was more snow cover in the Daviess and Henderson counties than in Caldwell county. Keep in mind that overwintering survival is just one of the variables that will, in part, determine the potential for SWCB problems in 2001. Historically, the date of planting of individual fields has been a key variable contributing to the potential for late season SWCB damage. Typically, fields planted after May 10 have an increased potential for this type of damage. What we can conclude:
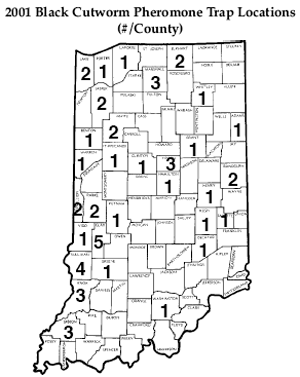
As Always, Black Cutworm Moths Are Beginning to Arrive– (John Obermeyer, Rich Edwards, and Larry Bledsoe)
Although at low numbers, black cutworm are beginning their movement into the Midwest from sites in south Texas and northern Mexico. Warm, moist air currents sweeping up from the Gulf Coast literally lifts these moths up into the upper atmosphere and carries them into Midwestern states. The direction that these weather systems track and the number of moths that are carried within them will determine whether they are brought into our area and whether or not they will pose a threat to our crops. As in the past, we will be trapping for this insect throughout the state (see “2001 Black Cutworm Pheromone Trap Locations” below). As we approach the more critical times for moth activity, that being later April and beyond, we will be watching for what we refer to as an “intense capture.” This is when 9 or more moths are caught in a trap over a two-day period. When and if this occurs, we will begin accumulating heat units (HU base 50°F) to determine when the first cutting of corn by the larvae should occur. This occurs approximately 300 HU after the intense capture. Watch for this information and Bug Scout’s alerts in future Pest&Crop’s Weather Update.
Black Cutworm Adult Pheromone Trap Report
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Wild Garlic, Wild Onion, and Star-of-Bethlehem- (Thomas N. Jordan)
Several samples and telephone calls have been received in the last two years about these weeds growing in no-tilled fields. They are members of the lily family, and thus are not controlled by any of the grass herbicides even though they look like a grass. All three are perennial species that like to grow in undisturbed areas, which make them ideal for no-tilled fields. Wild onion is the least common species found in crops, but it is usually the most common of the three found in lawns. Particularly in the southern part of the state, star-of-Bethlehem can be a problem in lawns and other areas such as golf fairways and parks. While wild garlic has traditionally been the most common species found in fields in the state, star-of-Bethlehem is the one that seems to be increasing the most in the last few years. This is probably due to one of two factors. One, it is spreading in no-tilled fields, or two, most people can already identify wild garlic but are not sure about the garlic looking weed they find increasing in their field. Star-of-Bethlehem at first looks like wild garlic, but upon close examination, you will see that the leaves are flattened with usually a light green to white midvein, and there is no garlic smell to the plant or the bulbs. It will bloom in mid-April to mid-May and have a single six-petal, white flower (like a star) on the top of the stalk. This plant, unlike wild garlic, will die back to the ground by mid-season and will not grow again until late fall or early spring of the next year. All three species are tolerant to normal use rates of glyphosate, and wild garlic and star-of-Bethlehem are tolerant to 2,4-D. Wild onion is controlled with 2,4-D. In wheat, Harmony Extra is excellent on wild garlic, but star-of-Bethlehem is somewhat tolerant to this herbicide. Harmony Extra is labeled for early preplant to soybeans and will control wild garlic but not star-of-Bethlehem. However, there is a 45-day waiting period before planting soybeans, so it is too late to use it this year. Canopy XL can be used as a burndown for wild garlic, but is weak on star-of-Bethlehem. Unfortunately for no-till, all of these species respond to tillage, and under tilled conditions they will not survive.
Getting Off To A Good Start– (Thomas N. Jordan) Production input cost is a primary concern to Indiana producers this year. One of their main concerns is the price of weed control. Going after the cheapest products, while a good idea, works only if they are right for the weeds that are present in a field. There is no substitute for knowing the weed spectrum for each field and matching the herbicides for those weeds. There are few if any truly cheap herbicides left on the market. If only one species of weed escapes the herbicide program in populations high enough to cause economic problems to the crop, additional applications will be needed. This could cause the overall cost of the herbicide program to increase by 50 to 100%. If the field is to be no-tilled, then the burndown program must be complete. This can only be accomplished if the right burndown products are used at the correct rates and when the temperature is favorable for good kill. Changing the rates of one of the mixture components may be needed. If there are a large number of broadleaf weeds such as marestail, fleabane, prickly lettuce, etc., additional 2,4-D might be needed; if a large number of grass species are present, then a little more Roundup or Touchdown will help. If the weeds can be controlled with Gramoxone, maybe an additional half pint will help achieve complete burndown. Adding a little more product to the burndown is cheaper than having escapes or regrowth which may take an additional late application of a product at the full labeled rate. Likewise, making the application a few days later when the temperature is warmer will cause the herbicides to work better. The other option is to add a residual herbicide to the burndown spray. In corn, adding a product that contains atrazine in the burndown tank mix can improve the control of many of the hard-to control weeds in no-tilled fields. There are many residual soybean herbicides that can be mixed with burndown treatments to boost the performance of the herbicides and provide a clean seedbed to start the crop off right. A healthy uniform crop stand can do wonders toward competing with weeds, especially late emerging weeds. Getting the crop off to a good start will make any herbicide program work better and will best allow less than maximum rates of postemergence herbicides to perform. A healthy and uniform crop stand will also come closer to eliminating the need for an additional postemergence herbicide application to control late emerging weeds. Listed below are 10 tips that I feel will help make a successful weed control program, and at the same time keep the cost of the program at a minimum.
It's Never Too Early to be Concerned About Spray Drift- (Case R. Medlin)
April 1999
Since drift management is the mandatory topic in this year’s private pesticide applicator licensing classes, it seems logical to say, “drift is still a problem.” I’m sure we will battle the drift problem again this year as well, but perhaps with a little planning and taking a few preventative measures, we/you can keep drift problems to a minimum. Although there are some other things you can manage to reduce drift, below are a few things you may want to consider.
Managing Weed Problems in Alfalfa- (Case R. Medlin, and Steven D. Siegelin) Typically, poor alfalfa stand management (e.g., low soil pH conditions, inadequate fertility, poorly timed insect management or improper mowing) results in reduced growth or death of alfalfa plants, which leads to open areas and weak alfalfa stands which invite weed infestations. So the most effective weed management programs should begin with crop management practices that encourage a healthy alfalfa stand. Your local extension office has many resources available to assist in these management decisions. However, even in well-managed stands weed problems can occur. This week calls concerning weed control in alfalfa have increased. Most of the calls are relative to winter annual weed problems (i.e., chickweed, mustards, and winter grasses) that invaded fields last fall or this spring. Winter annual weed problems as well as summer weeds can reduce the yield, quality, and longevity of alfalfa stands. The winter annual weeds are noticed as problems during the first of April and until the first harvest. If you wait much longer, the most effective control practice is mowing since (1) the weed/crop competition has already occurred, (2) often those plants have already gone to seed, and (3) the plants are most likely too large to control with in-season herbicide products. However, while the alfalfa is still dormant and/or the weeds are still relatively small a number of herbicides are labeled for their control. Controlling Winter and Summer Annuals in Established AlfalfaPursuit® can be applied postemergence to dormant or actively growing alfalfa past the second trifoliate leaf stage, but is weak on some large winter annuals. If winter annuals are anticipated next year, Pursuit® can be applied in the fall to small emerging weeds or as a preemergence treatment before weed emergence. Pursuit® is also very effective for controlling many summer annual broadleaves and grasses that invade open areas left by winter annuals, but has a 30-day pre-harvest interval that may not fit with aggressive mowing schedules. Other herbicide alternatives include Sencor®, Lexone®, and Sinbar® for control of many broadleaf weeds but must be applied to dormant alfalfa. Poast®, Poast Plus®, and Select® can be used to control summer and winter annual grasses throughout the year. Weed Management in Preparation for Establishing an Alfalfa StandWeed management for a new alfalfa stand should begin well in advance of seeding. Perennial weeds (including bindweed, Canada thistle, curly dock, dandelion, johnsongrass, hemp dogbane, quackgrass, and others) are a primary concern since management options for controlling them in established alfalfa are limited. Most herbicides applied during alfalfa production do not effectively control the root systems of perennial weeds. Therefore perennial weeds should be intensively managed beginning one or two years before alfalfa establishment. If perennial weeds are a problem in an established stand, there are alternatives. Good alfalfa stand management (i.e., maintaining proper soil pH and fertility conditions, and timely insect management) coupled with repeated mowing over several years can effectively deplete the root reserve of perennials such as bindweed, Canada thistle, hemp dogbane, johnsongrass and milkweed. Although mowing schedules and mowing heights should be based on sound management practices, they should also help eliminate any potential spread of weed seed. Another alternative includes using spot treatments of nonselective herbicides such as Roundup Ultra Max®, or Touchdown®. The disadvantage of these treatments includes further death of the alfalfa stand, which leaves larger open areas for weeds to invade. Poast®, Poast Plus®, and Select® are labeled for perennial (i.e., johnsongrass, and quackgrass) as well as annual (i.e., foxtail, crabgrass, barnyardgrass, etc.) grasses in established alfalfa stands. Please refer to Tables 1, 2, and 3 for other herbicide alternatives for weed control in alfalfa. Table 1 Table 2 Table 3
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Growth Stages of Wheat– (Gregory Shaner)
[Note: The following article appeared in Pest & Crop last year, but it being reprinted here for more convenient reference.] For effective management of wheat, it is important to recognize the stages of growth as the crop develops. Heading date is a commonly-recognized indicator of relative maturity, but the ability to recognize other growth stages is important for judging the progress of the crop and making management decisions, such as application of herbicides or fungicides, and for predicting the consequences of disease or injury to the crop. Two scales are commonly used to record the growth stage of wheat: the Feekes scale and the Decimal (Zadoks) scale. The Feekes scale is older and has been used widely since the early 1950s. The Decimal scale is designed to make finer distinctions among stages of crop growth, and is probably used more in Europe than in the US, although pesticide labels in the US are starting to use both scales. In the Feekes scale, growth stages are divided into 11 major categories. Head emergence, flowering, and grain filling (Feekes Growth Stages 10 and 11) are further subdivided. The Feekes Growth Stage scale is presented below, with a description of crop development stage that corresponds to each number. Corresponding Decimal Growth Stages are given in brackets. There are nine major divisions (1-9) in the Decimal scale, with ten possible subdivisions (0-9) for each major division. For example, the tillering stage is denoted by 2 in the Decimal scale, and the second digit indicates the number of tillers per plant. The exact number of tillers per plant prior to jointing is more detail than usually required. The Feekes scale simply notes whether tillers have begun forming (FGS 2), or whether tillering is essentially completed (FGS 3), without requiring the counting of tillers. In winter wheat, the period from beginning of tillering to completion of tillering may extend for a considerable time, from autumn into the following spring. Likewise, the precise limits of FGS 4 and 5 are not clear. Depending on planting date, variety, and weather in the fall, plants may reach the pseudostem erection stage in the fall, or only in the spring as the crop comes out of dormancy. Jointing (FGS 6) is a more clearly determined stage of growth. The original Feekes scale simply defined stage 6 as when the first node was visible at the base of the shoot. The Decimal scale provides a more practical definition for this stage, namely when the distance between the crown and the first stem node is at least 1 cm (0.4 in.), and I have included this in the growth stage table. When the second aboveground node is at least 2 cm (0.8 in.) above the first node, the plant has reached FGS 7. The stage when the flag leaf first appears (FGS 8) is important for application of a foliar fungicide. Soft red winter wheats in Indiana typically have four aboveground nodes when fully developed. The sheath of the uppermost leaf (flag leaf, F) arises from the top node. The leaf below the flag leaf (F-1) arises from the next node down, etc. Thus, leaf F-3 arises from the lowest aboveground node. The lowest aboveground node is near the ground when it first appears, but will move upward somewhat as the stem elongates. At FGS 8, there are usually two clearly differentiated nodes on the stem. The lowest will average about 7 cm (2-3/4 in.) above the soil line. The second node (from which leaf F-2 arises) will be about 15 cm (6 in.) above ground. The third node will usually be visible, but because it is only about 1 cm (0.4 in.) above node 2, it is not counted. As the wheat continues to grow, the distance between nodes increases, and the fourth node becomes evident. Once the flag leaf blade has fully emerged, the flag leaf sheath extends. By this time, the head enclosed in this leaf sheath is swelling, and the plant enters the boot stage (FGS 10). The heads of all plants in a field will not emerge from the boot in exact synchrony. Stages 10.1 through 10.5 are best assigned according to when heads on about half the plants have reached the indicated degree of emergence. Flowering in wheat begins roughly in the middle of the head and progresses both upward and downward. Flowering at a given position in the head can be judged by the presence of extruded anthers. Ripening is judged by removing developing kernels from the center of several heads and determining whether the contents are watery, milky, or at the soft or hard dough stages. By the time wheat has reached FGS 8, leaves F-5 and below are usually withered, from infection by Septoria, Stagonospora, and other fungi. The next leaf up (F-4) usually withers about the time heads have fully emerged. In the absence of Septoria and Stagonospora blotches, powdery mildew, or other foliar diseases, leaves F-3 through F should remain green until the wheat approaches maturity. Often, however, disease destroys leaves at each layer of the canopy prematurely. Fungicide control is aimed at maintaining these leaves, particularly F and F-1, in a healthy condition. If a grower is planning to apply a fungicide at flag leaf emergence (FGS 8), it would be helpful to know when that stage will be reached, relative to some earlier, easily determined growth stage. The jointing (FGS 6) and 2-node (FGS 7) stages can be determined with precision if a wheat field is monitored frequently. The time required for a plant to progress from either of these stages to FGS 8 is not constant. It depends on weather conditions, particularly temperature. Over many years, we have monitored wheat crop development in various field trials, and the following observations can give some guidelines for the average and ranges of times required for plants to progress from one growth stage to another. For example, we have found that it takes about a week to progress from FGS 6 to FGS 7, and another 8 days to go from FGS 7 to FGS 8 (with a range of 5 to 10 days). It can take from 3 to 8 days for the flag leaf blade to fully expand (going from FGS 8 to FGS 9). It can take from 9 to 16 days to progress from FGS 9 to full head emergence (FGS 10.5) or the beginning of flowering (FGS 10.5.1).
Yellowing of Wheat– (Gregory Shaner)
Now that we have entered a period of fluctuating temperatures, wheat that has started to green up may start showing a pale green to yellow mosaic on young leaves. These symptoms can result from infection by either of two soilborne viruses: soilborne wheat mosaic virus and wheat spindle streak mosaic virus. Both viruses persist in a common soilborne fungus. This fungus infects wheat roots in the fall. Infection by the fungus itself is of little consequence, but it does allow transmission of the viruses to the plant. Symptoms of virus infection don’t appear until the following spring. The timing of symptom development depends on weather. A long, cool spring with intermittent periods of warm and cold weather seems to favor symptom development. We have not yet had any periods of really warm weather, but temperatures are predicted to climb into the 60s over the next few days. If this period of warm weather is followed by another period of cold weather, symptoms will probably appear. Both viruses cause a yellowing of foliage. Soilborne wheat mosaic virus, as its name implies, causes a mosaic – narrow, pale green to yellow, wavy-margined streaks on the leaf blade. Symptoms of wheat spindle streak mosaic virus infection are similar, but the streaks tend to taper at both ends, hence the name “spindle.” From a distance, fields or parts of fields have a pale green or yellow appearance, as though they are deficient in nitrogen. In practice, it is very difficult to distinguish these two diseases based on symptoms. Both viruses may be found in the same field, and both viruses may infect a single plant. Wheat spindle streak is reported to be more uniformly distributed throughout fields than is soilborne wheat mosaic virus. Most varieties of soft red winter wheat grown in Indiana have some degree of resistance to these viruses. They may show some yellowing during periods of fluctuating temperatures during the spring, but once the cold weather is past, these varieties tend to outgrow the symptoms on lower leaves and there is probably little damage. A few varieties are more susceptible. The intensity of yellowing is greater, and is accompanied by stunting, reduced tillering, and death of some plants in the field. These varieties will suffer economic damage from these diseases. There is no remedial action that can be taken at this stage. If a variety develops severe symptoms, don’t plant it again next year. There are plenty of varieties with good resistance.
Soybean Sudden Death Syndrome– (Gregory Shaner and Scott Abney)
Sudden death syndrome is a mid summer disease, so why worry about it now? A number of factors determine whether sudden death syndrome will be a problem, and one of these is planting date. Soybeans that are planted early are at greater risk of sudden death syndrome than soybeans planted later. The majority of severe sudden death syndrome samples submitted to us by soybean growers have been from early-planted fields. Other risk factors include the susceptibility of the soybean variety planted, weather, and how often soybeans are grown in a field. A soilborne fungus, Fusarium solani f. sp. glycines, causes sudden death syndrome. Soilborne fungi don’t move around quickly, so the disease may be severe in one field, and mild in another field nearby. The difference in severity of disease from one field to another is primarily the result of differences in the abundance of the fungus in the soil. Fields that had severe sudden death syndrome last year probably have high populations of the fungus. Given that much of Indiana’s soybeans are now planted in a 2-year rotation sequence with corn, most fields that were afflicted with sudden death syndrome during 1998 and 2000 will probably be planted to corn this year. Does this mean the risk of sudden death syndrome is less for soybeans planted this year in fields that were in corn last year? Not necessarily. F. solani f. sp. glycines is ubiquitous in soybean fields, and there is now no way to avoid the threat of sudden death syndrome. The fungus survives in soil and can persist for several years even though soybeans are not grown. The key to reducing loss from this disease is through managing it, not avoiding it. There are two things a grower can do to reduce the risk of sudden death syndrome: avoid early planting and use a variety with some degree of resistance. At present, there are only a few varieties with proven, reliable resistance to sudden death syndrome. Those that do appear to have resistance are not totally resistant, but they are damaged less and will yield more than susceptible varieties in the presence of the disease. Work done by us (USDA-ARS and Dept. of Botany and Plant Pathology) has shown that resistant varieties yield as well as susceptible varieties in the absence of the disease, so there appears to be no yield penalty associated with resistance. Often when choosing a variety, a farmer will say that yield is the major factor, and disease resistance is only a secondary consideration. Actual yield and disease resistance are not unrelated issues. The only reason we worry about diseases in crops is because they reduce yield and quality. A variety may have excellent yield potential, but if it is susceptible to a disease such as sudden death syndrome and the disease develops, then that yield potential becomes meaningless. Where soybeans are being planted in an area that has a history of sudden death syndrome, planting should be done during the period of May 5-20 and a variety with some degree of resistance should be used if possible. Research at Purdue and elsewhere has shown that there is no yield advantage to planting early. Early planting increases the risk not only of sudden death syndrome, but also of various seedling diseases that thrive under conditions adverse for soybean growth.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Will Starter Fertilizer Increase Corn Yield?– (Robert Hoeft, Univ. of Illinois and Greg Willoughby, Purdue's Crop Diagnostic Training and Research Center) As planting season begins to get under way in southern Indiana and as we are getting itchy to start in the northern part of the state with the good weather we have been having, our minds are drifting to our planters. Our planter is one the critical machines we often take for granted especially the fertilizer system. A few thoughts from Dr. Hoeft and myself on starter fertilizer that you should consider, particularly in no-till/zone till systems, to protect your yields. To settle some debate, starter fertilizer can result in increased early-season growth, but this early-season growth does not always translate to increased yield. What it does is increases the health (or stamina) of the plant so that it can better handle future yield robbing stresses. In addition, the stronger plants will be able to react more favorably to conditions that may offer a positive yield response when they occur. The magnitude of starter effects are most likely to be seen under two conditions.
In summary, as you contemplate putting the planter in the ground make sure that you have taken into account the benefit of the fertilizer system. As always, check for proper calibration to ensure accurate placement and the proper amount of product to maximize the efficiency of your inputs.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Transgenics and Cross-Pollination- A problem?– (Rich Edwards) The recent ruling in Canada against a farmer who claimed he did not knowingly plant Monsanto’s Roundup Ready® canola and that transgenic pollen likely blew into his field, thus contaminating his crop, made me think back on a study that a graduate student of mine, Bob Abrams, a colleague, and I did back in 1976. The results of this study were published in 1978*. No, we were not working on transgenics back then. In fact, none of us could spell the word, much less define it. What we were looking at were yields and cross-pollination of soybeans as affected primarily by bees. Bob was a bee buff and had chased them from Indiana to Nepal and back. The only experience I had had with them was a chance contact with a swarm that decided to pass through my car window near the Amana Colonies in Iowa during my graduate school days at Iowa State University. That car was never quite the same, and I never did get the driver’s seat cleaned up! No, I never thought I’d pull that publication out of the file again. Sorry Bob! This feeling did not have anything to do with the job Bob did on his Master’s thesis and subsequent publication because he really did a nice job. One just normally does not go back to these without good reason and there never was a reason until now. But the concern over pollen from transgenic crops moving to non-transgenic ones made me think about this work and drove me to the dusty publication archives in the Purdue Extension Entomology Office. What I rediscovered was a darn good publication by a then young man who had distinguished himself earlier in his career by publishing his undergraduate senior thesis at the University of Michigan on prostitution and urban culture in the 1890’s. I’m not sure how he got from prostitution to entomology, but there are some insect critters that have certainly benefited from the world’s oldest profession and may have provided the link. What Bob’s work did do was address cross-pollination in a crop like soybeans and thus focus on one of the central issues in the debate over the problems with transgenics – contamination through cross-pollination. When most of us academic-types think about soybean and pollination, we think about a plant that is self-pollinated. Therefore, under most conditions there should be very little cross-pollination. The soybean flower pretty much keeps the pollination process in-house, and thus the soybean is a good candidate for production as a transgenic with little worry about field-to-field contamination. However, there is something missing from the above statement. What about those critters that move about within and among soybean fields looking for something to feed upon, whether it be another insect or some plant part. And if that movement takes that critter to the flower of the soybean plant during pollination, there is the opportunity for it to pick up pollen and move it about that field and surrounding fields depending on its foraging habits and ability to move significant distances. One of the things we wanted to study in Bob’s work was whether or not critters like bees can cross-pollinate plants and, if they can, to what degree. If we could get high enough levels of cross-pollination, we were thinking about the possibility of producing hybrid soybean. What we found was a substantial amount of cross-pollination taking place due to insects moving among fields. We observed 27 species of insects visiting soybean flowers during the blooming period with the minute pirate bug, Orius insidiosus (Say), and the honey bee, Apis mellifera L, being the most numerous. Bob noted on numerous occasions that honeybees were seen flying up an down soybean rows after the soybean foliage had covered the rows. Hybridization rates of as high as 18-20% were noted within some test areas of fields, with 3-7% being the norm. Fairly significant cross-pollination I’d say for a self-pollinated plant! Do our findings from 1976 have significant ramifications in regard to the development and use of soybean transgenics in 2001 and beyond? Perhaps! The bottom line is that contamination of one field by pollen moving into it from another field is a real possibility and needs to be addressed. Whether or not this means much when it comes to potentially compromising the Order of Nature has yet to be seen and/or thoroughly studied. However, based on my experiences dealing with Mother Nature and this “old” study, I suggest caution! Perhaps “terminator” technology, or whatever you want to call it, is not such a bad idea if transgenics are here to stay! If not, well that’s another story and back into that dusty file goes the publication. Sorry again Bob! *Abrams, R. I., C. R. Edwards, and T. Harris. 1978. Yields and cross-pollination of soybeans as affected by honey bees and alfalfa leafcutting bees. American Bee Journal. Vol. 118 (8), 555,556, 558, 560.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ARS Releases New Insect-Resistant Corn Germplasma Line (March 29, 2001, USDA ARS News Service) Agricultural Research Service scientists recently released a new corn germplasm line that will be a source for developing corn plants resistant to the southwestern corn borer, Diatraea grandiosella, and the fall armyworm, Spodoptera frugiperda. ARS researchers at the Crop Science Research Laboratory in Mississippi State, Miss., led by plant geneticist William P. Williams, have developed the corn germplasm line, Mp716, that is resistant to leaf feeding by these formidable pests. The new line was developed by self- pollinating a cross between two other germplasm lines for eight generations and then selecting for the desired traits. The milky-white larva of the southwestern corn borer appears in early June throughout much of the South. After feeding on the whorl—tightly coiled leaves within the stalk—of the corn plant, it moves down the stalk and begins to tunnel within. If the larva feeds on the bud of the plant within, the whorl, the plant’s entire yield is lost. Female southwestern corn borers can lay from 300 to 400 eggs in their lifetime. Agriculture Extension specialists in Mississippi estimate that this pest produces about $1 million dollars worth of damage annually in that state alone. The fall armyworm attacks corn and a variety of other crops including tomato, cotton and alfalfa. Like the southwestern corn borer, this pest also damages the whorl of the plant. This feeding produces frayed holes in the leaves that become apparent when they are unfurled. In addition, the larvae of the fall armyworm also feed on immature ears and tassels. The new germplasm line was evaluated for three years by infesting plants in the whorl stage of growth with 30 young larvae and checking for damage 14 days later. Mp716 was found to be only moderately damaged by these insects. The genetic material for this new germplasm line will be deposited in the National Plant Germplasm System where it will be made available for research purposes.
Manure Locator Site – (Todd Applegate, Purdue University, Animal Sciences)
Myself and others in the Dept. of Animal Sciences have been working on a manure locator web-site (or as NRCS has affectionately coined it a manure ‘dating’ service). Producers can advertise manure type/amounts on the site for free. As such, the primary objective of this site is to help producers get the word out about the benefits of ‘organic’ fertilizers at a time when inorganics are rather expensive. Other links that are available on the site are links to manure testing labs & custom applicators. If you know of any additional persons that are not listed on these pages, feel free to contact me with that information and we will add them to the list.
The site can be found at: <http://www.ansc.purdue.edu/ManureLocator>. In the months to come, we will also be adding lists of certified crop advisors, among other links. If you have any suggestions on any additional information that may be of benefit to you or your producers, please let us know. As you talk with folks in your counties, please let them know about this resource.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||