Pest & Crop Newsletter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Economic Alfalfa Weevil Damage Heading Northward– (John Obermeyer, Rich Edwards, and Larry Bledsoe)
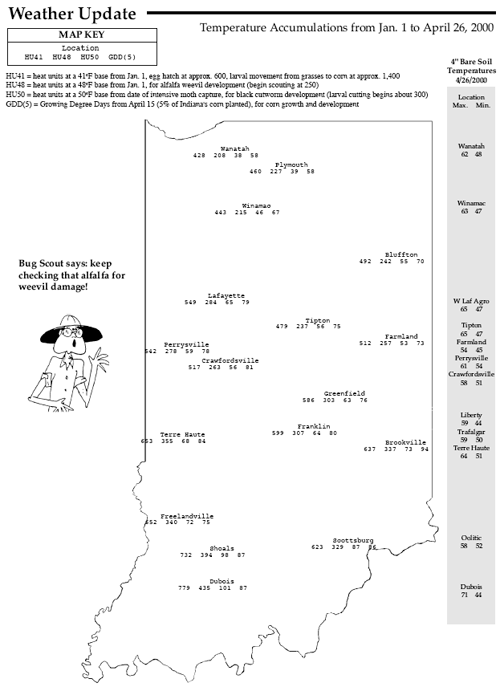
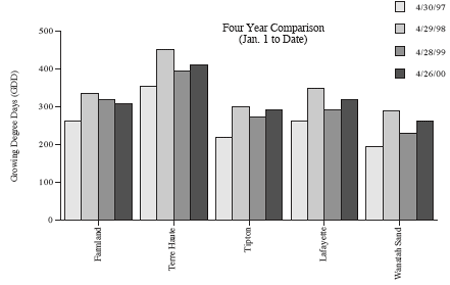
Surveys of west central and northwestern Indiana alfalfa fields this past week (see “Alfalfa Weevil Larval Survey”) show that alfalfa weevil feeding continues at high levels. The number of plants with tip feeding has reached as high as 100% (range 40 to 100%) with up to 7.4 weevil larvae per stem (range 1.7 to 7.4). Producers in all areas of northern Indiana should be evaluating their alfalfa fields, if they have not already done so, as soon as possible. However, controls should not be applied until thresholds are reached. Refer to the following management guidelines for northern Indiana and the heat units (base 48°F) in the “Weather Update.” See Pest&Crop #3 for scouting procedures and issue #5 for recommended control materials.
Correction to Insecticide Table for Alfalfa Weevil– (John Obermeyer) Please omit methyl parathion (Penncap-M) from last week’s (Pest&Crop #5) “Insecticides for Alfalfa Weevil Larval Control” table. Though we always try to provide you accurate information, especially with pesticides, we do make mistakes. Remember, the product label is the law, always check the label before using the product!
Early Emerging Soybeans Attract Bean Leaf Beetle– (John Obermeyer, Rich Edwards, and Larry Bledsoe)
A small percentage of soybeans have been planted and are beginning to emerge. Overwintering bean leaf beetle become active on warm days in the spiring and seek legumes, e.g., alfalfa and soybean, to feed and propagate. Large numbers of bean leaf beetle are possible this spring because of the last fall’s population and the mild winter. One of the most critical times for soybean damage is from emergence through the establishment of the first trifoliolate. If cotyledons are being destroyed before the unifoliolate leaves fully emerge or if the growing point is severely damaged, reduced yields are likely. However, once trifoliolate leaves have unrolled, soybean can tolerate up to about 40% defoliation without yield loss. For cotyledon, and unifoliolate stage soybean, refer to the following thresholds*:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Wheat Streak Mosaic Epidemic– (Don Hershman and Doug Johnson, University of Kentucky) The following article is being reprinted with permission from Kentucky Pest News, April 24, 2000. Last week we started to receive samples and phone calls which suggested that wheat streak mosaic had reared its ugly head again in Kentucky. The last and only recorded epidemic of this virus disease in KY was in 1988. Current information indicates that the greatest incidence of wheat streak mosaic is across the southern portion of the state, extending from east of Bowling Green all the way to Fulton County. However, the full extent of the epidemic has yet to be determined. It is likely that additional counties will report the disease as crop development continues. Wheat streak mosaic virus is transmitted by the wheat curl mite. This association and why we believe that wheat streak is a problem this spring will be addressed later in this article. Symptoms: Severely diseased plants are fairly distinctive. They will be severely stunted; leaves will be small and will have a “spiked” appearance. Lower leaves and tillers may turn brown and die. Plants may have a flaccid look about them and may look like they are having difficulty standing. Leaves will show extensive yellow streaking, especially from the middle of leaves towards the tips. The typical discontinuous yellow streaks in leaves may be so extensive that leaves will have a yellow, “bleached out”appearance. Some leaves will have a mosaic symptom. Severe symptoms, such as described above, indicate that infection occurred in the fall or early winter. The prognosis for severely disease plants is not very good and 75% or greater yield loss can be expected. Fields have large percentages of severely diseased plants are candidates for destruction and replanting to alternate crops. In some cases, only portions of fields may severely diseased. In those instances, it may only be necessary to destroy a portion of those fields. Although, currently, many fields do have a high percentage of severely diseased plants, many other fields are just now beginning to show yellowing and streaking, but are not stunted. If symptom expression occurs early, such as prior to flag leaf emergence, then expect those plants to deteriorate rapidly. However, if plants are in the boot stage or beyond when symptoms begin to be expressed, then expect only moderate to light yield effects. In fact, based on information provided to us by Kansas State University Extension Plant Pathologist, Dr. Bob Bowden, most plants which show late symptoms will have reduced test weight, but that is about it. These fields are certainly NOT candidates for destruction, but they may also not be good candidates for adding additional inputs, such as foliar fungicides or insecticides. Basically, the impact of late-appearing wheat streak mosaic symptoms depends upon the incidence of diseased plants in a field. Obviously, the greater the number of diseased plants, the more impact there will be on crop test weight and, thus, profitability. So now to the key questions. Why is wheat streak severe this year? Could it have been prevented? What can be done to reduce future occurrence? To answer these questions we need to look at how the pathogen, wheat streak mosaic virus, gets into wheat. Certainly we would like to have a clear idea of why we have this problem After all, it has been 12 years since a wheat streak mosaic epidemic has occurred in Kentucky. So, lets see if we can construct a usable model to provide an explication for this event. A complete life history of the mite is given in Entfact:117 - Wheat Streak Mosaic Virus and the Wheat Curl Mite. It is available from your county agent or from the Entomology Web site at: <http://www.uky.edu/Agriculture/Entomology/enthp.htm>
What is different about this growing season? We probably have the mite, the virus, and alternate hosts here every year but we rarely have an epidemic. On Tuesday April 18, 2000, Dr. Hershman and I met with several county extension agents, producers, consultants, farm store managers and technical representatives. These “meetings” took place in infected fields in and around Logan and Warren Counties. We were able to view the severity and distribution of affected fields, and perhaps more importantly gather background information from people involved in local wheat production. While we are not able to solve the problem, we do think that between all of us we can now explain what has happened. Wheat is the ideal host. The mite can live on some other grasses but it does not do well. Since we do not ordinarily have the problem, our wild hosts are not sufficient to provide an epidemic. In looking at the literature and talking with people from Kansas, they always see the problem when wheat from a previous cropping season is allowed to live continually through to the next wheat season. This can happen in several ways. Wheat from shattered heads at harvest will germinate in the field and then may grow continuously tho the next planting. Wheat may live continuously in abandoned fields and of course from spillage along roads etc. The point is whether in or near production fields, in problem areas wheat is growing throughout the time between crop, allowing the mites and virus to survive, move about and increase in number. This is the green bridge. However, our production system is quite different from those out west. We usually have several months (during the summer) between wheat harvest and planting when wheat is generally not present. So how do we get this Green Bridge? And what was particular to the1999-2000 season? The answer to this question became very apparent by talking with the local individuals. Because of the drought stress of the summer of 1999, the soybean crop (especially double crop) did very poorly. As a result producers were unwilling to provide the additional weed control that normally would have controlled volunteer wheat and other grasses in the soybean crop. As a result, the area currently suffering from Wheat Streak Mosaic had quite a lot of volunteer wheat. Hence our green bridge! This is most likely the core of the problem. However, the long warm fall and very mild winter certainly allowed the mites to remain active for much of the growing season. Once everyone was aware of how the mite/virus movement and survival controlled the disease epidemic cycle, it was easy to find examples of where the green bridge had occurred. This does not explain every individual case, but it certainly does explain the core reasons for why we are seeing this epidemic in this year, in this area. One of the more difficult questions is why do we find fields that are very evenly infested and do not appear to be near areas of volunteer wheat? My best explanation for that is to use scattered rain storms as a model. Once these mites get into the air they may simply “rain” down on an area. This movement would probably be of longer distance, provide a rather even dispersal on a local level, and be a matter of chance as to where they might settle out. You might get rained on and you might not. So what is to be done? It appears that this pest is very difficult to detect and it cannot be controlled with insecticides. The only real remedy is to stop the formation of the green bridge. If we return to normal production practices this problem may take care of itself. If however, it appears that fields are going to be abandoned or if other cropping practices evolve that allow volunteer wheat to prosper, we will have to do something to control the volunteer wheat. This will have to be an area wide effort. Just doing the right thing on your own farm will not be enough. Everyone needs to understand that wheat growing during the off season is a threat to commercial production. Without the cooperation of a large number of individuals working in wheat production, we probably would not have been able to sort out this puzzle.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Calculating Heat Units for Corn Phenology– (Bob Nielsen)
Heat units are simply a way to measure the accumulation of heat over some period of time. Different methods exist for calculating heat units depending on a) the crop or biological organism of interest and b) the whim or personal preference of the researcher. The calculation method most commonly used for determining heat unit accumulation relative to corn phenology is the formula first suggested by the National Oceanic and Atmospheric Administration in 1969 and termed the ‘Modified Growing Degree Day’ formula in 1971. This method calculates daily accumulation of GDDs as the average daily temperature minus 50°F. The ‘modification’ refers to the limits imposed on the daily maximum and minimum temperatures allowed in the calculation. Daily maximums greater than 86 degrees F are set to equal 86 in the calculation of the daily average temperature. Similarly, daily minimums less than 50°F are set to equal 50 in the calculation. Example 1: If the daily maximum temperature was 80°F and the minimum was 55°F, the GDD accumulation for the day would be ((80 + 55)/2) – 50 or 17.5 GDDs. Example 2: (Illustrating the limit on daily maximums): If the daily maximum temperature was 90°F and the minimum was 72°F, the GDD accumulation for the day would be ((86 + 72)/2) – 50 or 29 GDDs. Example 3: (Illustrating the limit on daily minimums): If the daily maximum temperature was 68°F and the minimum was 41°F, the GDD accumulation for the day would be ((68 + 50)/2) – 50 or 9 GDDs. Obviously, the ability to calculate daily heat unit accumulations is dependent on your having access to daily maximum and minimum temperatures. If you do not have your own max/min recording thermometer, you can guesstimate the daily highs and lows by recording the temperatures shortly after sunrise (approximate daily low) and late in the afternoon (approximate daily high). Temperature data for specific locations are also recorded and reported at the Indiana Climate Page on the Web at <http://shadow.agry.purdue.edu/sc.index.html>. Don’t forget, this and other timely information about corn can be viewed at the Chat ‘n Chew Café on the World Wide Web at <http://www.kingcorn.org/chatchew.htm> . For other information about corn, take a look at the Corn Growers’ Guidebook on the World Wide Web at <http://www.kingcorn.org/index.html>.
Leaf Staging Methods for Corn– (Bob Nielsen)
Growth staging in corn can be confusing, some even claim it’s a shell game. The confusion exists because there is no universally accepted method to stage corn. Agronomists have their method, hail insurance adjusters have theirs, and herbicide labels are in between the two. Leaf Collar Method. Agronomists determine leaf stage in corn by counting the number of leaves on a plant that have visible leaf collars, including the first short rounded-tip leaf. The leaf collar is the light-colored ‘band’ visible at the base of an exposed leaf blade. “Droopy Leaf” Method. Hail insurance adjusters also begin counting with the first short leaf, but continue counting beyond the uppermost visible collar to that leaf which is 40 to 50 percent exposed. In knee-high corn or older, the tip of this leaf is typically also pointing downward, hence the name “droopy leaf” method. The few post-emergence corn herbicide labels that refer to corn leaf stage basically use the ‘droopy leaf’ method, except that the first short leaf is not counted. Leaf Collar vs. Droopy Leaf. Up to the 5- to 6-leaf collar stage, the ‘leaf collar’ method will typically result in a leaf stage that is one less than the ‘droopy leaf’ method. After corn reaches 18 to 24 inches in height, the ‘leaf collar’ staging method will typically result in a leaf stage that is two less than the ‘droopy leaf’ method. As corn plants develop, the lower few leaves typically die or are ripped from the stem by expansion of the stalk or by developing roots. Consequently, growth stage identification can be difficult on older plants when you aren’t sure which leaves to begin counting first. But, not to worry, growth staging can still be accomplished! First, dig or pull a plant without breaking the stalk. With a knife, split the stalk down the middle, completely through the root ball. Look for the first noticeable internode (the whitish area between the ‘woody’ horizontal stalk nodes) above the triangular ‘woody’ base of the stalk. This internode’s length is typically only 1/2 to 3/4 inch. Carefully determine which leaf’s sheath attaches to the node immediately above the first noticeable internode. This leaf is usually Leaf #5. Once Leaf #5 is identified, then stage the plant by counting the remainder of the leaves up the plant that have visible leaf collars. If you are wanting to determine growth stage in order to use a defoliation/yield loss chart, then remember to add 2 to that number to equal the ‘droopy leaf’ method.
Predicting Corn Phenology for Phun and Profit– (Bob Nielsen)
Phenology: A branch of science dealing with the relations between climate and periodic biological phenomena (Webster’s New Collegiate Dictionary, 1973) Predicting when a field of corn should reach particular leaf stages can be useful for planning certain post-emergence applications of herbicides and sidedress N fertilizer, especially if your farming operation is so large that field inspections are difficult to work into your busy schedule. Recent research by one of my former graduate students helps fine-tune our ability to predict the portion of corn phenology that we call leaf stage development. Review of Concepts. For a refresher on how corn leaf staging is done, be sure to read the accompanying article on “Leaf Staging Methods for Corn”. Another topic that probably needs reviewing is the concept of heat units (HUs) or growing degree days (GDDs). The concept is important because corn phenology is very dependent on temperature. Heat unit or GDD calculation for corn phenology is also described in an accompanying article. Predicting Corn Phenology. Given an understanding of corn leaf stage development and heat unit calculation, you are now prepared to predict what leaf stage of development a particular field is at given its planting date and temperatures since planting. It is useful if you know when the crop emerged, but if not you can estimate that event also. Corn emergence typically requires 100 to 150 GDDs.
Based on my former graduate student’s research (Kirby Wuethrich. 1997. Vegetative and Reproductive Phenology of Fourteen Hybrids of Dent Corn (Zea mays L.). Purdue Univ.), we now know that corn leaf developmental rates can accurately be described in two phases. From emergence to leaf stage V10 (ten visible leaf collars), leaf emergence occurs approximately every 85 GDDs. From leaf stage V10 to the final leaf, leaf emergence occurs more rapidly at approximately one leaf every 50 GDDs. Example 1: A field was planted on April 28 and emerged on May 5. Since May 5, approximately 535 GDDs have accumulated. Based on our research data, the estimated leaf stage for the crop (without looking at the field, mind you) would be between V6 (6 leaves x 85 GDDs = 510 GDDs) and V7 (7 leaves x 85 GDDs = 595 GDDs). Example 2: A field was planted on April 28, but you do not know exactly when it emerged. Since planting, approximately 785 GDDs have accumulated. If you assume that the crop emerged in about 125 GDDs, then the estimated leaf stage for the crop would be between V7 and V8. The estimation stems from first subtracting 125 from 785 to account for emergence, then dividing the result (660) by 85 to equal 7.8. Example 3: A field was planted on April 28 and emerged on May 5. Since May 5, approximately 1200 GDDs have accumulated. Your experience with these calculations tells you that the crop is likely beyond V10 (equal to 10 x 85 or 850 GDDs). First subtract 850 from 1200 (knowing the crop is at least at V10). Divide the result (350) by 50 to equal 7 additional leaves for a total estimated leaf stage of V17. Keep in mind that estimates of leaf stage development are only that, estimates. One of the factors that most influences the accuracy of these estimates is the existence or not of growth-limiting stresses. However, what I have described here will put you in the proverbial ballpark in determining which fields are at which leaf stages.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Know Your Friends: Bigeyed Bugs– (Bob Wright, University of Kentucky) The bigeyed bugs, Geocoris spp., are generalist predators commonly found in a variety of crops and noncultivated plants nationwide. The common name refers to a characteristic feature of these insects, their rather large, protruding eyes. Bigeyed bugs are members of the family Lygaeidae (seed bugs), but belong to a subfamily containing only predaceous insects. There are many different Geocoris species found throughout the U. S. The adults range in color from black and white to tan. The nymphs resemble miniature grayish adults. They are small (3-4 mm long) insects capable of feeding on a variety of types of prey such as insect eggs, aphids, mealybugs, spider mites, leafhoppers, bugs, and small caterpillars or beetle larvae. Each nymph may consume up to 1600 spider mites during its immature stages, and as many as 80 mites a day as an adult. Other laboratory studies have shown that each G. punctipes nymph consumes about 250 soybean looper eggs before it reaches the adult stage. In yet another lab study adult bigeyed bugs consumed up to four Lygus bug eggs per day. In laboratory and field studies they have been observed to feed on dead insects, although when given a choice they prefer live prey. Geocoris also feed on plants; maximal survival and reproduction in the laboratory occurs when they have a mix of plant and insect food. In the laboratory they can survive for months on sunflower seeds and water. This plant feeding behavior is probably related to their evolution from seed feeding lygaeid bugs, and allows them to survive periods of low insect abundance. Their omnivorous feeding habits allow them to survive in a variety of habitats and contribute to biological control of pest species that become abundant. There is no evidence that their plant feeding causes significant injury to the plant. However, their plant feeding makes them susceptible to systemic insecticides; studies in cotton have shown reduced Geocoris survival on plants treated with systemic soil insecticides. Bigeyed bugs have piercing sucking mouthparts. Field observations indicate that Geocoris usually attack prey simply by walking or running up to a potential prey, extending their beak and quickly inserting it into the prey. Geocoris may lift prey into the air, preventing the prey from attempting escape by running. They may drop to the ground if disturbed.
Like all true bugs, they go through incomplete metamorphosis. The immature nymphal stages resemble the adults, except they lack functional wings. Nymphal stages have similar behavior and feeding habits as adults, but tend to feed on smaller prey. Most bigeyed bugs go through five nymphal stages. Under laboratory conditions nymphal development takes about 30 days at 25xC and 60 days at 20xC. Although not well studied, it is reported that bigeyed bugs overwinter as adults or as eggs depending on the species and location. Bigeyed bugs are particularly common in cotton, peanut, and soybean fields, and seem to prefer weedy areas in gardens. They are also common in managed turfgrass, such as home lawns. Bigeyed bugs were effective predators of corn earworm eggs in cotton in experimental mass releases, but are not available for sale commercially. However, recent studies by USDA-ARS scientists have identified new artificial diets for bigeyed bugs based on ground beef and beef liver. This diet costs $2.50 per pound, compared with $300 a pound for rearing with insect eggs. Bigeyed bugs reared on the new diets produced more offspring, often mature faster and are up to 50% larger than field collected insects. Although not commercially available yet, these predators are important in natural control in many agricultural settings. Midwest Biological Control News - Volume IV, Number 12 December 1997
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||