Pest & Crop Newsletter
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Rootworms Feeding on Soybean Roots?– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
Some observant pest managers have seen what appears to be rootworm larvae feeding on soybean roots. They look, move, feel, and feed like rootworms, but they are NOT rootworms! These are the larvae of the bean leaf beetle that were so abundant early this spring. The overwintering beetles, after feeding and mating, laid eggs in the soil which produced these larvae. The bean leaf beetle larva is white and distinctly segmented, with a brown head and brown hardened area at the posterior end of its body. To the naked eye, it looks identical to the corn rootworm larva. It feeds on soybean nitrogen fixing nodules and, to a lesser degree soybean roots. This damage has not been shown to significantly reduce yields.
Bean leaf beetle pupae, which also look like rootworm pupa, are being found in the soil with these larvae. This indicates that the first-generation adults will soon be or are already emerging throughout the state. These beetles will be found feeding on soybean leaves throughout most of July, during that time they will once again lay eggs in the soil producing larvae and pupae. This will give rise to the second generation adults, that once emerged, will feed on soybean foliage and pods until the crop is mature. Once cold temperatures set-in, the beetles will then find a suitable overwintering site for next spring. There are some questions about the bean leaf beetle being a carrier and vector of a viral disease, bean pod mottle virus. Symptoms of bean pod mottle virus may resemble injury from herbicide drift or symptoms of soybean mosaic virus. Infected soybean plants may have mottled or crinkled leaves and plants may be stunted. Plants may also show green stem syndrome, wherein stems remain green after pods have matured. This is not a new disease, it has been recognized in the southern states for years (first identified in 1958). By 1974, this virus was identified in Arkansas, Illinois, Iowa, Louisiana, North Carolina, South Carolina, Mississippi, and Virginia. Why the recent excitement? Certainly the numerous beetles feeding this spring had seed companies contemplating treatment thresholds and the possible added benefit to controlling them to prevent this disease. The symptoms may also help explain, or be an excuse for, some of those mysterious “herbicide drifts” in recent years. Perhaps newer soybean cultivars are more susceptible than in years past. Perhaps with time and research there will be a better answer to this insect/disease relationship. An excellent introductory discussion about this disease is available in Iowa State’s April 24 newsletter Integrated Pest Management which can be seen on the web at <http://www.ipm.iastate.edu/ipm/icm/2000/4-24-2000/newsoyvir.html>.
Grasshoppers in Field Crops– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
Ron Blackwell, IPM Surveyor, has observed grasshopper nymphs feeding in field edges. Though the damage was minor at this time this could quickly change as the grasshopper mature.
Eggs, laid last fall in the soil, normally hatch from late May through July. After the nymphs hatch, they normally feed for 2 to 3 weeks near the area where the eggs were laid. When their food source becomes scarce or when their early feeding sites are mowed or otherwise destroyed, the nymphs move to other feeding sites, including nearby crops. The best time to manage threatening grasshopper populations is when they are still in the nymphal stage (wingless). They are easier to control at this time since they are less mobile and more susceptible to the insecticide. However, you should not apply a control just because they are in the nymphal stage. An economic population needs to be present to justify treatment. Another consideration is that under high moisture conditions, a naturally occurring fungal disease may quickly spread through the grasshopper population. Grasshoppers seen clinging to crops and weeds, and not hopping away as you approach, are most likely diseased and will soon die. To determine the need for grasshopper control, check crop and non crop areas to pinpoint infestations. In at least 5 random locations within each infested area, estimate the number of grasshoppers within approximately one square yard. Determine the infestation level for each sample area, and for each crop and non crop area as a whole. Treatment of field margins is probably justified if counts exceed 15 or more nymphs or 8 adults (winged) per square yard. In soybean fields, control may be needed if defoliation levels exceed 40% prior to bloom or 15% from blooming to pod fill. In corn, it may be advisable to treat if an average of 3 or more grasshopper nymphs per square yard are counted. In many cases, spot treatment may be sufficient to control the grasshoppers.
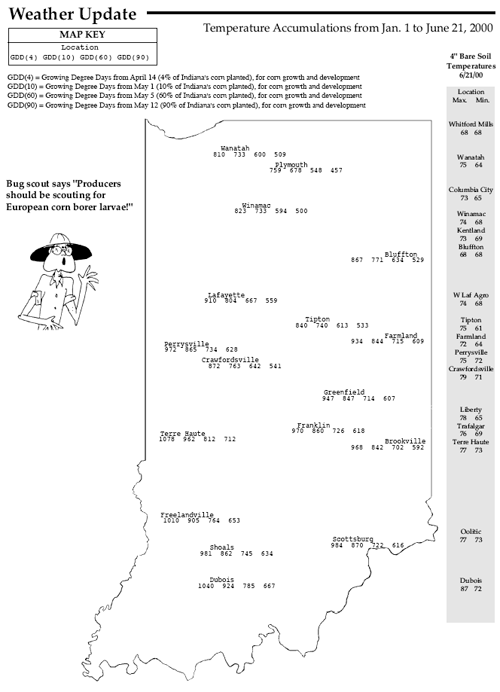
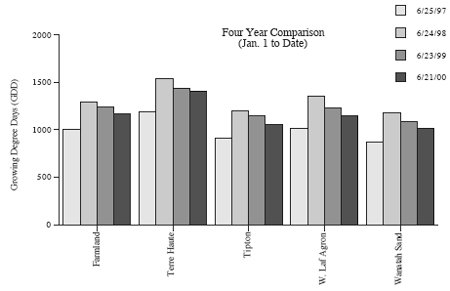
Although Corn Borer Populations Are Generally Low, Some Fields Have Significant Damage– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
Random surveys by Ron Blackwell, IPM Surveyor, (see “European Corn Borer Survey”) and telephone calls to our office concerning European corn borers indicate that populations are generally low. However, populations in northeast Indiana are threatening in some fields. Surveys in Allen and DeKalb Counties on June 21st revealed corn borer “shot hole” feeding on 41 to 97 percent of the corn plants in fields surveyed. Obviously, producers in northeastern Indiana need to spend some time scouting their fields. While the first generation appears to be causing only minor concern in other areas of the state, producers should still examine their corn for borer activity and should use the management guidelines given in Extension Publication E-17, European Corn Borer in Field Corn (Rev. 3/00), before making control decisions. Shortly, many borers will be entering corn stalks. Once in the stalk, the borer is protected from insecticides. This has likely already occurred in southern Indiana. The “window of opportunity” for control may close within a few days of receiving this newsletter for northern Indiana producers. Therefore, producers should closely examine stalks for borer entry before making control decisions. If treatment is necessary, it is recommended that the control material be applied before 1/3 of the borers have entered the stalk. Midvein leaf feeding indicates that stalk tunneling is soon to begin; frass accumulation in a leaf axial signals that the stalk has been entered by a borer.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In last week’s newsletter I reported that common rust was already present in corn. It was possible to find several pustules on lower leaves of some hybrids, but severity was much greater in a seed corn field, southeast of Indianapolis, where I have some fungicide plots. During the past few days, rust has increased substantially in this inbred. As I mentioned in the article last week, the rust fungus does not overwinter in Indiana, but arrives each spring from areas to the south. Given that primary inoculum is exogenous, and the fact that rust is becoming severe in this one seed corn field, there is a good chance that it is becoming equally severe in other seed corn fields planted to susceptible inbreds. If fields have not already been scouted, they should be, to determine whether it is time to apply a fungicide.
Soybean Sudden Death Syndrome– (Gregory Shaner)
It is too early for sudden death syndrome to appear in soybeans, and it is not possible to say whether the disease will be a problem later this year. However, a couple of factors may be setting the stage for a problem this year. Like all plant diseases, an epidemic of sudden death syndrome requires three conditions: a widespread susceptible population of host plants, a widespread and virulent pathogen, and favorable environment. We had all these conditions during the summer of 1998, and there was a widespread and severe epidemic of sudden death syndrome in Indiana and neighboring states. There is no high degree of resistance to sudden death syndrome and it was evident during 1998 that many varieties were susceptible. That situation has probably not changed dramatically since then. So, it is reasonable to believe that once again we have a generally susceptible population of soybeans. It was likewise evident in 1998 that the pathogen, Fusarium solani f.sp. glycines, was widely distributed in Indiana soybean fields. Many of the fields that had soybeans in 1998 will have soybeans during 2000, given that a great many soybean acres are in a 2-year rotation with corn. As for the environment, cool, wet seasons seem to favor disease outbreaks. Most important seems to be heavy rainfall that saturates soil when pods are just starting to develop. The fungus infects lateral roots early in the season, and invades the taproot. Saturated soils during early pod development apparently trigger the production by the fungus of a translocatable toxin that moves into foliage and produces the characteristic foliar symptoms. Upper leaves of infected plants initially show an interveinal chlorosis. These areas rapidly turn brown and then the leaf blades shrivel and drop off. Leaf petioles remain attached to the plant, giving it a skeletal appearance. If the taproot and lower stem are split open longitudinally, the cortex will be discolored, but the pith will be white. If an infected plant is dug up when the soil surface is wet, it may be possible to see patches of blue pigment on the surface of the taproot. These are masses of spores of the fungus. The blue color fades quickly in dry air. The disease is more of a problem in beans that are planted early, and is rarely seen in double-crop soybeans. A lot of soybeans were planted early this year, and although conditions were initially dry, many areas of the state have had frequent rains during the past 3 weeks. Temperatures have been cool until recently. If rains persist for another couple of weeks, as pods start to develop, we may see sudden death syndrome again this year. It not possible to say whether sudden death syndrome will be a problem, but as discussed above, the early planting of many soybean fields this year, and the weather we have had so far may have set the stage for a problem, depending on weather conditions during the next 2 or 3 weeks.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ear Size Determination in Corn– (Bob Nielsen)
Potential ear size is an important factor that contributes to the grain yield potential of a corn plant. Severe plant stress may limit the potential ear size, and thus grain yield potential, before pollination has even occurred. Optimum growing conditions set the stage for maximum ear size potential and exceptional grain yields at harvest time. Ear size determination begins by the time a corn plant has reached knee-high and finishes 7 to 10 days prior to silk emergence.
Ear Shoot Development. There are as many potential ears as there are leaves on the plant since every stalk node has an axillary bud associated with it. However, while axillary buds exist at the upper 6 to 8 nodes of the stalk, they normally never become active. Careful dissection of stalks at about growth stage V10 (10 leaf collars) will reveal 8 to 10 ear shoots. Each ear shoot is attached at a stalk node, behind its respective leaf sheath. At growth stage V10, the identifiable ear shoots are composed primarily of husk leaf tissue. The developing ears themselves are only a fraction of an inch in length. Initially, the lower ear shoots are longer than the upper ones because the lower ones form first. Later on, the upper one or two ear shoots take priority over the others and become the harvestable ears. Brace root development will also rip off ear shoots at the lowest stalk nodes. The uppermost, harvestable ear will normally be located at the 12th to 14th leaf. Development of the upper ear is favored over the lower ones because of hormonal ‘checks and balances’, plus the proximity of the upper ear to the actively photosynthesizing leaves. Damage to the upper ear prior to pollination can allow one or more of the lower ones to develop into harvestable ears. Ear Size Determination. Total kernel number is determined by the number of kernel rows and the number of kernels per row. Row number is one of several yield components in corn. Every pair of rows is generally equal to 20 bushels per acre (for average populations and ear lengths). Kernel number per row is another yield component of corn. For a 16-row ear, one kernel per row is equal to about five bushels per acre (for average populations). Typically, from 750 to 1000 ovules (potential kernels) develop on each ear shoot. Actual (harvestable) kernel number per ear averages between 400 and 600. Kernel row number determination is complete by growth stage V12. Kernel rows initiate as ‘ridges’ of cells that eventually differentiate into pairs of rows. Thus, row number on ears of corn is always even unless some sort of stress disrupts the developmental process. True row number is often difficult to visualize in tiny ears dissected from plants younger than about the 12-leaf stage. Row number is determined strongly by plant genetics rather than by environment. This means that row number for any given hybrid will be quite similar from year to year, regardless of growing conditions. Exceptions include... Deep row cultivation after growth stage V8 may prune root systems severely enough to hinder row number determination. Applications of certain sulfonylurea herbicides beyond the labels’ stated growth stage restrictions can decrease yield potential by interfering with final row number determination on the upper portion of the ear. Nearly complete defoliation by hail prior to growth stage V12 may photosynthetically ‘shock’ the plant and limit row number determination. The potential number of kernels per row is complete by about 1 week before silk emergence. Kernel number (ear length) is strongly affected by environmental stresses. This means that ear length will vary dramatically from year to year as growing conditions vary. Severe stress can greatly reduce potential kernel number per row. Conversely, excellent growing conditions can encourage unusually high potential kernel number.
Flooding & Ponding: How Long Can Corn Tolerate 'Wet Feet'?– (Bob Nielsen)
Recent intense rainfall events (technically referred to as ‘toad stranglers’ or ‘goose drownders’) have caused flooding or ponding of corn fields in low-lying areas throughout the state. Other fields, while not technically flooded, have nonetheless been saturated for lengthy periods of time. The oxygen supply in the soil is depleted after about 48 hours in a flooded soil. Without oxygen, the plant cannot perform critical life sustaining functions; e.g., nutrient and water uptake is impaired, root growth is inhibited, etc. How long can corn tolerate ‘wet feet’ before dying? If it survives, what other consequences of ‘wet feet’ can develop? The growth stage of a corn crop greatly influences whether ponding or saturated soils kills, severely stunts or mildly stunts the corn plants. Plants younger than V6 are susceptible to damage for two reasons. First of all, the growing point in such young corn is at or below the soil surface and therefore is also subject to the stress of oxygen-depleted conditions. Once the growing point is above the water level the likelihood for survival improves greatly. Secondly, plants younger than V6 are in the process of trying to successfully establish a vigorous root system. Stunting or death of roots by oxygen-depletion can be a major stress for a plant that is not yet fully established. Prior to leaf stage V6 (six-leaf stage as measured by visible leaf collars), corn can survive only two to four days of flooded conditions. If temperatures are warm during flooding (greater than 77F) such young plants may not survive 24 hours. Cooler temperatures prolong survival. If flooding in corn is less than 48 hours, crop injury should be limited. To confirm plant survival, check the color of the growing point (it should be white and cream colored, while a darkening or softening usually precedes plant death) and look for new leaf growth three to five days after water drains from the field. Plants older than V6 will tolerate ponding or saturated soils longer for essentially the opposite reasons. As plants develop beyond V6, rapid stalk elongation elevates the growing point region above the soil surface and, thus, away from the direct stress of flooded soils. Secondly, an older crop’s root system will simply be larger and consequently the crop can tolerate a certain amount of root death without dying or dramatic stunting. Nonetheless, extended periods of saturated soils plus warm temperatures will take their toll on the overall vigor of the crop. Some root death will occur and new root growth will be stunted until the soil dries to acceptable moisture contents. As a result, plants may be subject to greater injury during a subsequently dry summer because root systems are not sufficiently developed to access available subsoil water. Concomitant (I found a new word in the dictionary!) with the direct stress of saturated soils on a corn crop, flooding and ponding can also cause losses of soil nitrogen through denitrification and leaching of nitrate N. Significant loss of soil N will result in nitrogen deficiencies in the corn crop that may cause additional yield loss. The Purdue reference cited below offers guidelines in estimating the amount of nitrogen loss due to saturated soils and making decisions on application of additional nitrogen fertilizer to fields once ponded. Certain disease problems which may become greater risks due to flooding and cool temperatures are corn smut and crazy top. The fungus that causes crazy top depends on saturated soil conditions to infect corn seedlings. There is limited hybrid resistance to these diseases and predicting damage is difficult until later in the growing season. Other flooding/ponding on-line references: Hail and Flooding Damage in Corn (Univ. of Illinois, 1999) Effects of Flooding and Ponding on Corn (AGF-118-95)(Ohio State University) Corn survival in wet conditions (Iowa State Univ.,1999) Predicting Early Season N Loss (Purdue Univ., 1998) Don’t forget, this and other timely information about corn can be viewed at the Chat ‘n Chew Café on the World Wide Web at <http://www.kingcorn.org/chatchew.htm>. For other information about corn, take a look at the Corn Growers’ Guidebook on the World Wide Web at <http://www.kingcorn.org/>.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Artificial Habitats Boost Predator Populations in Soybean - (Midwest Biological Control News, Volume VII, Number 1 May 2000) Predation by generalist predators, such as spiders and ground beetles, can reduce pest damage in many crops. Providing temporary refugia during cyclical farming disturbances has been used in China for more than 2000 years to encourage spiders, but this technique has not been systematically investigated on a larger scale in western-style agriculture. After conventional tillage of soybeans, populations of spiders and their egg sacs normally decline by up to 75%. When modular habitats, consisting of chicken wire loosely filled with bedding straw, were placed in tilled fields, there were 5 to 36 times more spiders compared with open fields, and spider egg sacs were 18 to 87 times more abundant. Spider diversity was also much higher, with 60% more species found in refugia than in the open field. There were also more harvestmen, ground beetles, and rove beetles when refugia habitats were provided. Throughout the season the microhabitat within the refugia is more favorable for survival of predators than in just soybean plants or weeds. For example, in early October, harvestmen were 90 times more abundant in refugia than in senescing soybeans. The refugia supply not only a place to live, but may also enhance the predators’ food base. Small arthropods feeding primarily on fungal hyphae and detritis present in the decaying straw, especially Collembola (springtails), were available as alternate prey. Spider populations peaked earlier in the growing season in refugia, at a time when herbivore control may be more critical for plants. Soybean seedlings grown within a yard of these modular habitats had 33% less damage than plants in areas without habitats (although this did not significantly increase soybean yield). Using temporary habitats can conserve naturally occurring predators to enhance pest control. Providing special habitats, such as the chicken wire cages used in this research, would only be feasible on a small scale, but mechanized distribution of discrete straw patches in a field after tillage or harvest operations may offer some benefit on a larger scale. Source:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||