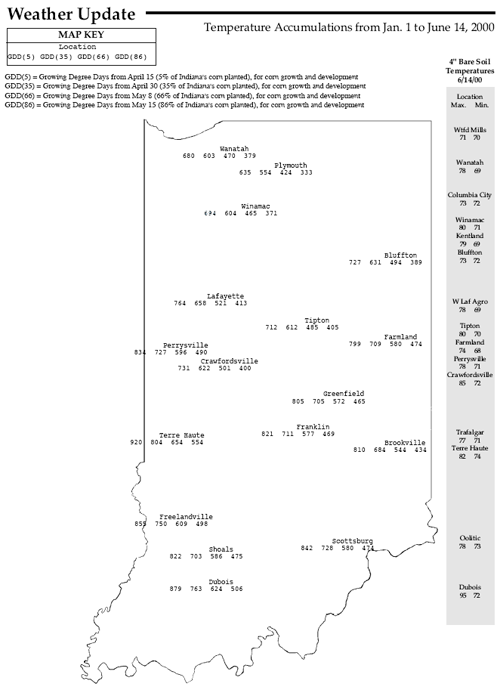
Pest & Crop Newsletter
|
|||||||
Corn Borer Still Low– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
Discussions with agribusiness personnel over the last couple weeks and black light catches have confirmed our suspicions about the first generation European corn borer … it is low through most of the state! Northeastern Indiana is the only area that has reported some evening windshield splatter, which has been confirmed with the black light catches. Recent storms, that have blessed many areas with much needed rain, have also helped reduce populations by drowning and dislodging small larvae attempting to establish in the corn whorls. This follows many days of hot conditions that too are detrimental to newly hatched larvae. Though corn borer is down, don’t count it out for the 2000 growing season. Even a small number of moths from this first generation, given ideal environmental conditions during mating and egg laying, can cause significant damage to some fields later this summer. Actively pollinating corn is most attractive to the moths for egg laying, usually this correlates to late planted fields. Stay tuned to future issues of the Pest&Crop as we track the moth flight and infestations of this pest throughout the season.
Suspect Rootworm Damage? Inspect For It Now– (John Obermeyer, Larry Bledsoe, and Rich Edwards)
At the Purdue Diagnostic and Training Center, we have been observing many rootworms feeding on corn roots. Until recent high temperatures, their development was slow. Now it is not difficult to find the creamy-white larvae in the root zone. Also, we have watched progression of feeding by the larvae, first on the primary root system then the nodal roots. Soon the brace roots, as they come in contact with the soil, will be fed on. If nodal and/or brace roots are pruned, plant lodging becomes likely, which leads to significant yield losses. If sampling reveals that rootworms are present in economically damaging levels and a cultivator or high-boy is still possible to get through the field, a rootworm insecticide should be applied immediately.
Using a shovel to sample, lift out the root mass and surrounding soil (about a 7” cube) and place on a dark surface (black plastic garbage bags work well). Carefully break up the clods and sort through the soil. Look for 1/4 to 1/2 inch long, slender, creamy-white larvae with a brownish-black head and tail. Once the soil has been separated from the root mass, inspect it for root scarring and pruning. You may find the rootworms under the leaf collars that are in close proximity to nodal roots, tear these leaves away to check. Also, you may even observe the rootworms’ hind-ends sticking out of roots. Repeat this process with several plants representing different areas of a field. An average of two or more larvae per plant represents a rootworm population that signals the need for a cultivation application. Insecticides applied after planting must be directed toward the base of plants. It is also important to cultivate the soil near the plants to incorporate the insecticide. Throwing soil up around plants will also promote the establishment of brace roots. A good brace root system will help prevent plant lodging and reduce losses due to rootworm feeding. If a no-till field has an economic population of larvae, placing the insecticide on top of the ground will normally not be effective. The only exceptions might be if the soil insecticide is watered in through irrigation or rainfall (ideally a 1/2” or more). Two liquid soil insecticides, Furadan 4F and Lorsban 4E, are labeled for post-directed applications. Furadan, being more soluble, would better move into the root zone if rainfall is minimal. If one decides to mix the insecticide with a liquid nitrogen source for a carrier, compatibility checks should be made. If you observe significant rootworm feeding and/or plant lodging in first-year corn, please contact us: (765) 494-4563 or (obe@purdue.edu). Thanks!
|
|||||||
Stewart's Wilt– (Gregory Shaner)
It does not appear to be a major problem, but Stewart’s wilt has been confirmed in at least a few Indiana corn fields. Stewart’s wilt is caused by a bacterium (Erwinia stewartii). The pathogen enters corn plants through wounds made by the corn flea beetle when it feeds on leaves of corn. Lesions are typically long and in early stages of disease development it is often possible to see the feeding tracks of the corn flea beetle within the lesion. At first lesions are pale green, but they rapidly turn brown. Margins are wavy. Infection that occurs early in the season can lead to rapid wilt of susceptible plants. Most field corn hybrids are fairly resistant to Stewart’s wilt and will show only localized lesions on leaves fed upon by flea beetles that harbor the bacteria. Inbreds and sweet corn hybrids are generally more susceptible to Stewart’s wilt and may develop a systemic infection that leads to plant death. In such plants, the bacteria move from the leaf originally infected into the growing point. Leaves that subsequently emerge will have pale green streaks, which rapidly turn brown. Then these leaves will wilt. A systemically infected plant may die well before tasseling or may hang on but fail to produce a viable tassel or ear. A few field corn hybrids are susceptible to Stewart’s wilt and will show systemic infection and wilting. Plants that are not systemically infected may develop leaf blight later in the season. When flea beetles feed on upper leaves, the same kinds of lesions described above may develop around the feeding sites. These long lesions may superficially resemble lesions of northern corn leaf blight. This phase of the disease is not as damaging as the wilt phase, but if blighting is extensive there may be reduction in yield and a predisposition to stalk rot later in the season. The likelihood of Stewart’s wilt is related to how mild the winter is. The corn flea beetle not only serves to spread the bacteria during the growing season, but also provides a means of overwintering. Corn flea beetles overwinter as adults, and those that acquired the bacteria before the onset of winter will carry the bacteria through the winter. In a warm winter, such as we had this year, more beetles, and therefore more Stewart’s wilt bacteria, will survive. Resistance is the most effective control, but for corn that is susceptible, insecticides may be effective by reducing vector feeding.
I have seen rust in a seed corn field. The pathogen is known as Puccinia sorghi, but don’t be fooled by the name. This rust fungus does not infect sorghum. Pustules were fairly numerous on leaves 6 and 7, and one or two pustules could be seen on leaf 9. The rust fungus does not overwinter in Indiana and therefore spores of the fungus must be carried north each spring by winds from tropical and subtropical areas. Spores settle out in still air or are scrubbed out of the air by rain. Those that land on corn leaves can infect if leaves are wet with dew or rain for as few as 6 hours. Primary infection from exogenous inoculum is never heavy enough to cause damage, but does establish the fungus in a field. It takes only a few days for an infection to mature into a uredinium (pustule). Each uredinium is a mass of spores, and each of these spores is capable of causing a new infection. When leaves are regularly wet at night, rust can increase explosively. The presence of rust in one field suggests that it may already be established in many other fields.
|
|||||||
Agronomist Fooled by Disease– (Bob Nielsen)
The story you are about to read is true. The name of the agronomist involved has been changed to protect the dumb. The moral of this story is to not wear blinders into a field while making crop problem diagnoses. In one field, ‘Bob’ discovered that 5 to 10 percent of the plants were essentially dead ‘from the neck up’, meaning that the first two or three leaves were okay but the remainder of the whorl was dead. A smaller percentage of the plants in the field were technically alive but deformed in their growth. An occasional plant showed one or more series of ‘shotholes’ in the leaves. Leaves of some of the affected plants displayed necrotic streaks along their length. Being a wise and experienced agronomist, ‘Bob’ knew that the whorl damage and death was often caused by insect damage to or near the growing point area of a young plant. Sure enough, upon splitting the stalks of the affected plants ‘Bob’ discovered that the growing points were not in good condition. “Ahah! I knew it!”, he exclaimed proudly to the farmer standing at his side looking on with amused interest. In fact, his first thought was of stalk borer because there appeared to be a feeding cavity where the growing point should have been. However, further inspection of the plants did not reveal any entry or exit wound that such an insect critter would leave behind. “Dang it!”, he said.
Just then ‘Bob’ remembered that several other Midwestern states had reported the occurrence of billbug injury in recent weeks. Now, ‘Bob’ had never seen billbug injury before but he knew that those pests somehow penetrated corn stems and injured meristematic areas with their snouts. Furthermore, the owner of the field was by now looking sternly over his shoulder wondering when he was going to make a concrete diagnosis. “Eureka!”, he exclaimed. “I betcha this injury was caused by billbug feeding!”, he confidently proclaimed and thereupon left the field. On the way home, ‘Bob’ called one of his insect buddies (John Obermeyer) at Purdue and proudly described his nice piece of detective work. John was skeptical of the diagnosis, partly because ‘Bob’ reported so few plants exhibited the characteristic ‘shot’ holes in the leaves that would result from either stinkbug or billbug injury to corn. He also thought the description of the cavity at the growing point area did not fit the diagnosis. Well, the chastened agronomist figured he would visit the field again and look for more evidence that would support either stinkbug or billbug injury and prove to John that the diagnosis was correct. So, back to the field on Monday for more investigative work. ‘Bob’ looked for evidence of nutsedge infestation because several references noted that the weed is a major host for billbug and is usually present when billbug injury occurs. None was found. He looked for billbug larvae in the stem or roots of the injured plants because several references noted that such things should be present. None were found. ‘Bob’ looked again for the characteristic ‘shot’ holes in the leaves of the affected plants, but only a small percent of the plants showed such holes. He looked for evidence of entry and exit wounds in the stems that could implicate either insect pest. Only a few plants exhibited such evidence. “Dang it!”, he said. The now thoroughly frustrated agronomist decided that it was time to ‘call in the calvary’ and so took a handful of injured plants back to the university’s Plant and Pest Diagnostic Laboratory. His friend John looked over the plants as did another colleague in plant pathology (Gail Ruhl). Lo and behold, the correct diagnosis of the injured plants turned out to be the wilt phase of Stewart’s Disease, that nasty bacterial disease transmitted by flea beetles. Gail identified the bacteria streaming out of the damaged tissue. I had never, I mean, uh..., ‘Bob’ had never seen the wilt phase of Stewart’s Disease before and, consequently, never considered that possibility when diagnosing wilted or dead whorls. Indeed, the wilt phase of this disease is fairly rare, particularly with commercial corn hybrids. Bottom Line: And now, ‘Bob’ realizes, the wilt phase of Stewart’s Disease can also be the culprit of wilted or dead whorls in young corn or deformed growth. Life-long learning: it’s not just a catchy phrase! Online Photo Gallery of this field at:<http://www.kingcorn.org/news/articles.00/Stew_Wilt-0614-Gallery.html> Online References for Stewart’s DiseaseStewart’s Disease (Iowa State Univ., 1995) Don’t forget, this and other timely information about corn can be viewed at the Chat ‘n Chew Café on the World Wide Web at <http://www.kingcorn.org/chatchew.htm>. For other information about corn, take a look at the Corn Growers’ Guidebook on the World Wide Web at <http://www.kingcorn.org/>
|
|||||||


 Japanese Beetles
Japanese Beetles