Pest & Crop Newsletter, Entomology Extension, Purdue University
- Stressed Corn, Pollination, and Beetles
- Western Bean Cutworm Moths Being Captured
- Foliar Fungicides and Insecticide Tank Mixes
- Black Light Trap Catch Report
![]()
Stressed Corn, Pollination, and Beetles - (Christian Krupke, John Obermeyer, and Larry Bledsoe)
- Large numbers of rootworm beetles being reported in some corn fields.
- Japanese beetle numbers are unequally distributed in areas/fields.
- Stressed corn and spotty pollination are making treatment decisions difficult.
- Length of silks and amount of pollen yet to fall is the key to fertilization.
Several calls have been received this week concerning the large numbers of rootworm beetles in cornfields. Many plants within these fields are being discovered with damaged roots from larval feeding. This, combined with uneven plant development, is resulting in spotty tassel and silk emergence. Treatment decisions are complicated as the beetles flock to a small percentage of pollinating plants.

Damaged silks still can receive pollen below the chewed area.
When fields are pollinating evenly, control may be necessary if the silks are clipped off to within 1\2 inch or less of the tip of the ear before 50% pollination is completed. Don’t judge the need for treatment on beetle numbers … SILK DAMAGE is the ONLY relevant factor for making treatment decisions. Producers have noted the beetles all over the plants, not necessarily feeding on the silks. Many rootworm beetles are in the corn whorls before tassels emerge, they are doing some economically insignificant but “showy” leaf feeding. As the tassels emerge, beetles may be seen feeding on the anthers and pollen. The high-protein pollen is their preferred food. They will never consume enough pollen to make a difference in pollination, however.
If you do decide to treat, remember that controlling beetles will only protect the silks that are still viable to receive pollen. Make certain that over half of the pollen hasn’t already been shed.
Before you decide to spray with insecticide, consider:
• Pollen is the rootworm beetles’ favorite food
• Japanese beetles prefer silks that are browning (completed)
• Many times more than enough pollen is shed to fertilize ears and feed beetles
• Japanese beetles tend to clump in a field, especially near the end rows
• Silks a 1/2 inch or longer can successfully receive pollen
• Silks can receive the pollen anywhere, not just the tip
For recommended control materials refer to Extension Publication E-219-W, Corn Insect Control Recommendations - 2007 (Revised 1/2007) <http://www.entm.purdue.edu/entomology/ext/targets/e-series/EseriesPDF/E-219.pdf>.
For related articles see the Chat ‘N Chew website at <http://www.agry.purdue.edu/ext/corn/cafe>.
![]()
Western Bean Cutworm Moths Being Captured - (Christian Krupke, John Obermeyer, and Larry Bledsoe)
- Early moth captures are higher than last year, still pale in comparison to western corn belt.
- Scouting in northwestern Indiana counties for egg masses/young larvae is advised in seed corn.
Like last year, pheromone trap cooperators are beginning to capture western bean cutworm moths. It is too early to speculate on this year’s flight intensity, but numbers so far are higher than last year. In addition, we have a larger number of trapping sites this year. You can track future Indiana, and other Midwestern state captures, via the Western Bean Cutworm Monitoring Network at <http://www.ent.iastate.edu/trap/westernbeancutworm/>
Some are already beginning to ask about scouting and treatment guidelines, especially in seed corn, for this pest. Obviously, since last year was the first observance of this insect in Indiana, we do not have experience or research to pull from. Instead, we look to the “experts”: University of Nebraska entomologists have known this sporadic pest for decades. They suggest that egg scouting begin once moths become active. In five different areas of a field, inspect 20 consecutive plants for egg masses which are laid on the upper surface of the top leaves of corn and/or larvae that may have hatched and crawled to the whorl and begun to feed. A treatment threshold of 8% of the plants with an egg mass and/or larvae in the whorl is suggested. Timely is critical, as soon as pollination begins, larvae will make their way into the ear via the silks and become impervious to insecticides.
One other important note: Please keep us informed of suspected larval infestations in the state. These observations and moth densities and range within Indiana will be reported in future issues of the Pest&Crop.

Western bean cutworm egg mass
![]()
Foliar Fungicides and Insecticide Tank Mixes - (Christian Krupke, John Obermeyer, and Larry Bledsoe)
The following article is being reprinted from Pest&Crop Issue #12, 2007. We desire for this message to be “heard.”
- Most insecticides do not control spider mites.
- Fungicides control both plant and insect pathogens.
- Dry weather + fungicide + insecticide = mite/aphid outbreak.
Fungicide applications, to both corn and soybean, are being planned for many fields throughout the state. Scouting to determine the need for a fungicide in the crop beforehand is unlikely, but there is a significant incentive to spray now and hope for yield boosts later. From the insect side, there are concerns associated with adding an insecticide to the spray mix just to “clean up” any insects that may be present.
Lurking in every corn and soybean field, all season long, are low numbers of various species of spider mites and aphids. We rarely notice these insects when conditions remain “typical”. However, treating fields with an insecticide may tip the balance in the favor of potential pest arthropods. This is because natural enemies recover more slowly from broad-spectrum insecticides compared with mites and aphids, which have an extremely rapid generation time.
Dry conditions exacerbate crop damage from mites and aphids. One major reason for this is because pathogens, that cause insect diseases, do not flourish. Just as crop diseases (most of which are moisture-loving fungi) are more likely during wet/high humid conditions, so are insect diseases. An epizootic is quite impressive, as potentially damaging populations of mites/aphids are quickly and thoroughly wiped out. Fungicides sprayed for crop diseases also control these beneficial pathogens. This is one reason why high-value crops, e.g., fruits and vegetables, receiving prophylactic (calendar sprays) of fungicide and insecticide often have spider mite flare-ups.
Lastly, it must be emphasized that insecticides do NOT increase yield, they only preserve it. Any yield-enhancing claims with insecticides are not only misleading but against the law. This one fact alone should make producers wary and monitor fields before treatments are made. Happy scouting and be informed!
![]()
Click here to view the
Black Light Trap Catch Report - (John Obermeyer)

Asiatic Dayflower Pretty, But Hard to Control (Glenn Nice and Bill Johnson)
Asiatic dayflower (Commelina communis) can be a problematic weed in lawns and gardens, but it appears over the past several years to be spreading into no-till corn and soybean. It is one of the 16 weeds mentioned in the Weed to Watch handout (if you would like a copy of this handout please contact Glenn Nice at Purdue University). As the common name would suggest, Asiatic dayflower was introduced to the US from Asia. We get occasional calls regarding Asiatic dayflower that was not controlled by glyphosate.
Asiatic dayflower is a member of the Commelinaceae or spiderwort family. This annual plant is a monocot with alternative lanceolate leaves that are 2 to 4 inches long. The base of each leaf clasps the stem. Asiatic dayflower has been misidentified as a grass with fleshy wide leaves. Although this might happen before it flowers, once it flowers, it shouldnt be mistaken for a grass. Asiatic dayflower has distinctive blue flowers approximately 0.5-1 inch wide with three petals. Flowers appear from June to October1. The flowers consist of three petals and three sepals, two larger petals above and one smaller below. The above two petals are blue and the one petal below appears faded blue or white2.

Asiatic dayflower
There are few herbicides that are effective on Asiatic dayflower. This is one of the weeds that is poorly controlled by glyphosate (Roundup Weathermax, Touchdown, Glyphomax, many). Iowa State University research reported that three applications of glyphosate (0.75 lb ae/A); at planting, 44 days after planting, and 63 days after planting was needed to achieve over 80% control. FirstRate® (cloransulam-methyl) plus Spartan® (sulfentrazone) also provided over 80% control3. The combination of these two products can also be found in Authority First®. In this study, Command® (clomazone), Basagran®(bentazon), Blazer® (acifluorfen), Cobra® (lactofen), Flexstar® (fomesafen), and Raptor® (imazamox) did not have much activity on this weed.
In cotton trials in Georgia, Stanley Culpepper reported that tank mixes of glyphosate with Cobra® (lactofen) or Valor® (flumioxazin) also provided 86 and 85% control respectively4.
Soybean:
The following recommendations are based on the limited information regarding Asiatic dayflowers control (see table below).
| Asiatic dayflower control recommendations | |||
| Herbicide | Active Ingredients | Rate | Comments |
| Authority First (FMC) | Sulfentrazone 62.1% Cloransulam-methyl 7.9% |
6.45 - 8 oz/A1 PRE | Do not apply on sand or soils less than 1% OM. apply at 2 to 6 leaf Asiatic dayflower |
| Spartan 4F (FMC) | Sulfentrazone 4 lb ai/gal | 6 - 10 fl oz/A1 | Will provide suppression |
| FirstRate (Dow Agrosciences) | Cloransulam-methyl 84% | 0.3 oz/A | Apply to 2 to 6 leaf Asiatic dayflower |
| Glyphosate + Cobra or Valor | Glyphosate (many) + Lactogen or flumioxazin | 1.1 lb ae/A + 12.5 fl oz/A or 2 - 3 oz/A1 | Apply before emergence or cracking if Valor is used. Apply to small Asiatic dayflower |
| 1Rate dependent on soil texture, pH, and organic matter | |||
1. Connecticut Botanical Society. Asiatic dayflower Commelina communis. Accessed June 29, 2007 <http://www.ct-botanical-society.org/galleries/commelinacomm.html>
2. L. Newcomb. 1977. Newcombs Wildflower Guide. Little, Brown and Company New York Boston. p 40.
3. B. Hartzler. 2003. Dayflower: a weed to watch? Accessed June 29, 2007 <http://www.weeds.iastate.edu/mgmt/2003/dayflower.shtml>
. 4. S. Culpepper. 2000. Crop Production Workshop. Mississippi State University.
![]()
Late-season Postemergence Issues in Roundup Ready Soybeans - (Article co-written by Mark Loux and Jeff Stachler, The Ohio State University, and Bill Johnson, Purdue University)
Weeds continue to defy control in a number of fields throughout Ohio and Indiana, and lambsquarters, giant ragweed, and marestail appear to top the list of most problematic weeds. No surprises here. Failure to make initial postemergence glyphosate applications early enough in the season can be a primary reason for poor control, due to the advanced size and age of weeds at the time of application. Dry weather caused many growers to delay postemergence applications, with the hope that an ensuing rain would improve conditions and the weeds’ response to herbicide. While the rain may have cooperated in some areas, the continued lack of rain in other areas resulted in older weeds that were generally more tolerant of herbicide. The effect of weather was evident in our research plots, where the effectiveness of the standard rate of glyphosate, 0.75 lbs ae/A, was reduced compared to wetter years, especially on lambsquarters and giant ragweed. Glyphosate was much more effective in our plots where it was applied at a rate of 1.5 lbs ae/A, or where the activity of preemergence herbicides resulted in smaller weeds at the time of postemergence application. Many growers continue to apply glyphosate at the rate of 0.75 lbs ae/A regardless of weed size and age or weather conditions, when applying a higher rate can be the better decision.
We are also hearing reports that many of the weeds not controlled by postemergence glyphosate applications have been infested with stem-boring insects. University research has shown some potential for a reduction in control when the stems have been infested with borers, and that increasing glyphosate rate can partially compensate for the reduction in control. This same research shows that borers are a problem primarily when the weeds were initially treated when they were too large or too old, which allows enough time for borers to infest the weeds. Applying glyphosate to small weeds, prior to the appearance of stem-boring insects, is the most effective means of avoiding this problem. We suggest that glyphosate be included in late-season applications even where stem borer is occurring, on the assumption that it will still contribute to the control of borer-infested weeds.
We can offer the following information on late-season control of weeds, especially those weeds that have escaped prior treatment.
Timing of Late Postemergence Applications
Our research generally shows that an interval of no more than 3 to 4 weeks should occur between postemergence glyphosate applications, which means that postemergence treatments should be largely finished by the second week of July. This allows enough time for surviving weeds to show some regrowth, but weeds have typically not regrown to a large size. It’s not necessary to allow the weeds to regrow to the point that they are taller than the soybeans. Weeds that are about the same height or slightly smaller than the soybeans can receive adequate amounts of herbicide spray. Soybeans treated in late July or later in the summer are more likely to be injured by glyphosate. Labels for Monsanto brand glyphosate products state that they can be applied to soybeans through the R2 growth stage (flowering), but not after the R3 growth stage begins. The R3 growth stage begins when one of the uppermost four nodes with a fully developed leaf has a pod that is at least 3/16 inches long. Application of glyphosate to soybeans that have started the R3 stage is not supported by the labels (i.e., not legal). We assume that the labels for most other glyphosate products contain similar guidelines, but we have not conducted a thorough search of labels to verify whether this is the case.
What to Spray?
It is important to keep in mind that the amount of glyphosate that can be applied to Roundup Ready soybeans between emergence and the end of R2 growth stage is 2.25 lb ae/A. So, if you applied 0.75 lb ae/A in the first postemergence treatment (22 oz/A of Roundup Weathermax or 24 oz/A of Touchdown, or 32 oz/A of a generic glyphosate), you can apply up to 1.5 lb ae/A in the second treatment. If you applied 1.5 lb ae/A in the first treatment, you are limited to 0.75 lb ae/A in the second treatment.
Marestail. Glyphosate-resistant marestail is widespread in southern Indiana and Ohio. However, most populations are somewhat suppressed by glyphosate, so the best strategy will be to apply the maximum amount of glyphosate allowed (0.75 to 1.5 lb ae/A) in a mixture with either FirstRate or Classic. There are a few populations of marestail that are resistant to both glyphosate and Classic/FirstRate, but Purdue surveys suggests that less than 10% of the populations in Indiana are resistant to both glyphosate and ALS inhibitors. FirstRate typically provides 5 to 10% better control of marestail than Classic. If you don’t know if whether your population is resistant to ALS inhibitors, consider past herbicide use in that field. Repeated use of FirstRate, Classic, or Synchrony in a field over the past 5 to 7 years increases the probability of ALS resistance.
Common lambsquarter. Under dry weather conditions, lambsquarter becomes more difficult to control with glyphosate or any other systemic herbicide. As mentioned above, the best strategy at this point in the season would be to increase the glyphosate rate if you have the ability to do so based on the rate used in earlier treatments. Including additional nonionic surfactant may result in slight improvements in lambsquarter control, but is not a substitute for increasing glyphosate rate where it is possible to do so. If the 1.5 lb ae/A rate of glyphosate was used in the first postemergence treatment, and you are limited to 0.75 lb ae/A, the addition of Harmony GT may improve activity on lambsquarter.
Giant ragweed. Populations that have evolved a low level of resistance to glyphosate can be found in at least 10 counties in Indiana and 11 counties in Ohio. The populations in Indiana are randomly scattered all over the state. The best strategy at this point would be to increase the glyphosate rate if you have the ability to do so based on the rate used in earlier treatments. If the 1.5 lb ae/A rate of glyphosate was used in the first postemergence treatment, and you are limited to 0.75 lb ae/A, the addition of FirstRate, Flexstar, Cobra, or Phoenix may improve activity. Important points to keep in mind as you think about tankmix partners for giant ragweed include:
1) Although we haven’t stumbled across a large number of populations resistant to both glyphosate and FirstRate, keep in mind that if you have previously observed poor performance by ALS inhibitors on giant ragweed in the field of concern, that the population may be ALS resistant. This could result in inadequate control with mixtures of FirstRate and glyphosate.
2) Flexstar carryover potential is greater when it is used late in the growing season. You may want to use Cobra or Phoenix instead, to reduce the potential for Flexstar carryover. Also, keep in mind that the use of PPO herbicides such as Flexstar or Cobra can antagonize the activity of glyphosate on some weeds. We have conducted trials to evaluate the use of different adjuvants in mixtures of glyphosate and Flexstar or Cobra, and our preliminary results suggest that we should stay with nonionic surfactant when mixing Flexstar and glyphosate. Our current research seems to indicate that while the use of crop oil concentrate reduces glyphosate activity, the mixture of Cobra plus glyphosate may be more effective when crop oil concentrate is added.
3) We have research in progress to resolve the rates that should be used in mixtures of glyphosate plus Cobra. While our information is incomplete at this time, this research seems to suggest that where the glyphosate rate is limited to 0.75 lbs ae/A in a late application, the Cobra rate should be 12.5 oz product/A. The Cobra rate can be reduced where it’s possible to use higher glyphosate rates.
Manganese and Glyphosate
The activity of glyphosate can be reduced when applied in mixtures with manganese, and this reduction in activity is of greater concern when trying to control weeds that have escaped prior herbicide treatment and under droughty conditions. Control of velvetleaf can be especially problematic when using these mixtures. Our best suggestion is to apply glyphosate and manganese separately when trying to clean up weedy fields this late in the season. Ideally, the glyphosate is applied first, followed by application of manganese at least one day later. When it’s impossible to avoid mixing glyphosate and managanese, follow these guidelines:
1. Use an EDTA-chelated form of managanese, which has less potential to reduce glyphosate’s activity in comparison with other forms of manganese.
2. Include ammonium sulfate in the spray mixture at the rate of 17 lbs/100 gallons. Add the ammonium sulfate to the spray tank first, followed by the manganese, followed by the glyphosate.
3. Use the highest glyphosate rate that can legally be applied, based on the amount used in previous postemergence applications (see comments on rate earlier in article).
![]()
Soybean Rust Fungicides - (Greg Shaner)
- Three fungicides have been added to the soybean rust arsenal.
When soybean rust first threatened North America, there were very few fungicides registered for use on soybean. To provide farmers with the means to control rust, states where soybean is produced applied to the US Environmental Protection Agency for exemptions to use several fungicides. The first exemptions were granted in time for the 2005 growing season. Recently, three additional fungicides were added to the list. These are Alto (cyproconazole), Caramba (metconazole), and Punch (flusilazole). All are in the triazole class of fungicides. Because none of the fungicides covered under a Section 18 Emergency Quarantine Exemption mention soybean rust on the label affixed to the container, users must have in their possession, when the fungicide is applied, the Section 18 label. This should be available from the dealer when the fungicide is purchased. These labels are also available, in pdf format, on Purdue’s soybean rust Web site. This can be accessed at <http://www.ppdl.purdue.edu/PPDL/soybean_rust.html>. This site includes labels for all fungicides available for use on soybean in Indiana under a Section 18 Emergency Quarantine Exemption.
The Section 18 exemptions are state-specific, so users in Indiana must be sure to have the label that is for Indiana. It is important to read the Section 18 label as well as the label that comes with the container carefully. Important information on the Section 18 label includes the time period for which the material can be used. After that period, unused product must be returned to the manufacturer or distributor. Other important pieces of information are use rates, pre-harvest restrictions (how late the fungicide can be applied), re-entry intervals, and crop rotation restrictions.
Soybean rust fungicide guidelines are also posted on the PPDL soybean rust site. This table and accompanying text provide guidance as to when to apply fungicides based on soybean growth stage and status of rust development, and what classes of fungicide to use.
Current status of rust can be found on the USDA PIPE web site, available at <http://www.sbrusa.net/>. Alternatively, call Purdue’s toll-free soybean rust hotline at 866-458-RUST (7878).

Silk Emergencee - (Bob Nielsen)
- Corn produces individual male and female flowers on the same plant.
- The ear represents the female flower of the corn plant.
- Severe soil moisture deficits can delay silk emergence and disrupt the synchrony of pollen shed and silk availability, resulting in poor kernel set.
As important as the process of pollination is to the determination of grain yield in corn, it is surprising how little some folks know about the details of cornfield sex. Rather than leaving you to learn about such things “in the streets”, take the time to read this article and the accompanying one on tassels and anthers (Nielsen, 2007b) that describe the ins and outs of this critical period of the corn plant’s life cycle.
The corn plant produces individual male and female flowers (a flowering habit called monoecious for you corny trivia fans.) Interestingly, both flowers are initially bisexual (aka “perfect”), but during the course of development the female components (gynoecia) of the male flowers and the male components (stamens) of the female flowers abort, resulting in tassel (male) and ear (female) development.
The silks that emerge from the ear shoot are the functional stigmas of the female flowers of a corn plant. Each silk connects to an individual ovule (potential kernel). A given silk must be pollinated in order for the ovule to be fertilized and develop into a kernel. Up to 1000 ovules typically form per ear, even though we typically harvest only 400 to 600 actual kernels per ear.
Technically, growth stage R1 (Ritchie et. al., 1993) for a given ear is defined when a single silk strand is visible from the tip of the husk. A field is defined as being at growth stage R1 when silks have emerged on at least 50 % of the plants.
Silk Elongation and Emergence
Silks begin to elongate from the ovules about 10 days prior to growth stage R1. Silk elongation begins first from the basal ovules of the cob, then proceeds sequentially up the ear. Similarly, silks from the basal (butt) portion of the ear typically emerge first from the husk, while the tip silks generally emerge last. Complete silk emergence from an ear generally occurs within four to eight days after the first silks appear.
As silks first emerge from the husk, they lengthen as much as 1.5 inches per day for the first day or two, but gradually slow over the next several days. Silk elongation occurs by expansion of existing cells, so elongation rate slows as more and more cells reach maximum size. Once pollinated, elongation of an individual silk will stop.
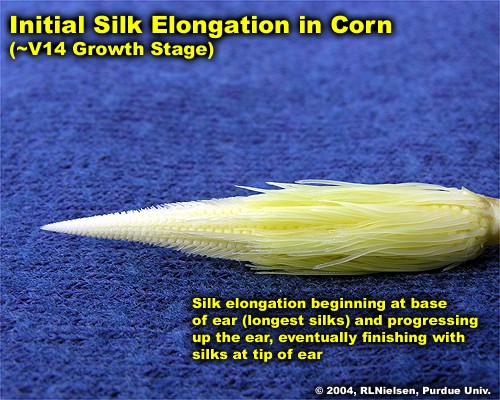

Silk elongation stops about 10 days after silk emergence, regardless of whether pollination occurs, due to senescence of the silk tissue. Unusually long silks can be a diagnostic symptom that the ear was not successfully pollinated.
Silks remain receptive to pollen grain germination up to 10 days after silk emergence, but to an ever-decreasing degree. Natural senescence of silk tissue over time results in collapsed tissue that restricts continued growth of the pollen tube. Silk emergence usually occurs in close synchrony with pollen shed, so that duration of silk receptivity is normally not a concern. Failure of silks to emerge in the first place, however, does not bode well for successful pollination.

Pollination and Fertilization
For those of you serious about semantics, let’s review two definitions relevant to sex in the cornfield. Pollination is the act of transferring the pollen grains to the silks by wind or insects. Fertilization is the union of the male gametes from the pollen with the female gametes from the ovule. Technically, pollination is almost always successful (i.e., the pollen reaches the silks), but unsuccessful fertilization (i.e., pollen tube failure, silk failure, pollen death) will fail to result in a kernel.
Pollen grain germination occurs within minutes after a pollen grain lands on a receptive silk. A pollen tube, containing the male genetic material, develops and grows inside the silk, and fertilizes the ovule within 24 hours. Pollen grains can land and germinate anywhere along the length of an exposed receptive silk. Many pollen grains may germinate on a receptive silk, but typically only one will successfully fertilize the ovule.
Silk Emergence Failure
Severe Drought Stress. The most common cause of incomplete silk emergence is severe drought stress. Silks have the greatest water content of any corn plant tissue and thus are most sensitive to moisture levels in the plant. Severe moisture deficits will slow silk elongation, causing a delay or failure of silks to emerge from the ear shoot. If the delay is long enough, pollen shed may be almost or completely finished before receptive silks are available; resulting in nearly blank or totally blank cobs. Severe drought stress accompanied by low relative humidity can also desiccate exposed silks and render them non-receptive to pollen germination.
The severity of drought stress required for significant silk emergence delay or desiccation can probably be characterized by severe leaf rolling that begins early in the morning and continues into the early evening hours. Such severe leaf rolling is often accompanied by a change in leaf color from “healthy” green to a grayish-tinged green that may eventually die and bleach to a straw color.
Silk Clipping by Insects. Although technically not defined as silk emergence failure, severe silk clipping by insects such as corn rootworm beetle or Japanese beetle nonetheless can interfere with the success of pollination by decreasing or eliminating viable or receptive exposed silk tissue. Fortunately, unless the beetle activity is nonstop for days, continued elongation of silks from the husk will expose undamaged and receptive silk tissue at the rate of about one inch or more per day.
Related References
Kling, Jennifer G. and Gregory Edmeades. 1997. Morphology and growth of maize. IITA/CIMMYT Research Guide 9. Int’l Institute of Tropical Agriculture. [On-Line}. Available at <http://www.iita.org/cms/details/trn_mat/irg9/irg9.htm> (URL verified 7/2/07).
Nielsen, R.L. (Bob). 2007a. A Fast & Accurate Pregnancy Test for Corn. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/EarShake.html>(URL verified 7/2/07).
Nielsen, R.L. (Bob). 2007b. Tassel Emergence & Pollen Shed. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/Tassels.html> (URL verified 7/2/07).
Ritchie, S.W., J.J. Hanway, and G.O. Benson. 1993. How a Corn Plant Develops. Iowa State Univ. Sp. Rpt. No. 48. [On-Line] Available at <http://www.extension.iastate.edu/pages/hancock/agriculture/corn/corn_develop
/CornPlantStages.html>. (URL verified 7/2/07).
Russell, W.A. and A.R. Hallauer. 1980. Corn. (a chapter in) Hybridization of Crop Plants. American Soc. of Agronomy-Crop Science Soc. of America. Madison, WI.
Steffey, Kevin. 2005. Rootworm Adults and Silk Clipping. Pest & Crop Bulletin, Univ. of Illinois. [On-Line]. Available at <http://www.ipm.uiuc.edu/bulletin/article.php?issueNumber=15&issueYear=
2005&articleNumber=2> (URL verified 7/4/07).
![]()
Tassel Emergence & Pollen Shed - (Bob Nielsen)
- Corn produces individual male and female flowers on the same plant.
- The tassel represents the male flower of the corn plant.
Over the next several weeks, the Indiana corn crop will move into the critical flowering stages of pollen shed and silk emergence. Success or failure during this period of the corn plant’s life will greatly influence the potential yield at harvest time.
As important as this process is to the determination of grain yield, it is surprising how little some folks know about the whole thing. Rather than leaving you to learn about such things “in the streets”, I’ve developed this article and the accompanying one on silking (Nielsen, 2007b) that describe the ins and outs of sex in the corn field.
Remember that corn has both male flowers and female flowers on the same plant (a flowering habit called monoecious for you trivia fans.) Interestingly, both flowers are initially bisexual (aka “perfect”), but during the course of development the female components (gynoecia) of the male flowers and the male components (stamens) of the female flowers abort, resulting in tassel (male) and ear (female) development.
Technically, growth stage VT occurs when the last branch of the tassel emerges from the whorl (Ritchie et. al., 1993). Portions of the tassel may be visible before the maximum leaf stage (final visible leaf collar) has occurred. Plant height is nearly at its maximum at growth stage VT. Pollen shed may begin before the tassel has completely emerged from the whorl.
The corn plant is most vulnerable to hail damage at growth stage VT since all of its leaves have emerged. Complete (100%) leaf loss at growth stage VT will usually result in complete (100%) yield loss by harvest. Even if pollination is successful, the ear shoots will usually die because few leaves remain to produce the necessary carbohydrates (by photosynthesis) to complete grain fill.
An individual tassel produces approximately 6,000 pollen-bearing anthers, although hybrids can vary greatly for this number. The anthers are those ‘thingamajigs’ that hang from the tassel during pollination. Under a magnifying lens, anthers look somewhat like the double barrel of a shotgun. Approximately 1,000 individual spikelets form on each tassel and each one bears two florets encased in two large glumes. Each floret contains three anthers. An anther and its attached filament comprise the stamen of the male flower.
As these florets mature, elongation of the filaments helps exsert the anthers from the glumes. Pollen is dispersed through pores that open at the tips of the anthers. Pollen shed usually begins in the mid-portion of the central tassel spike and then progresses upward, downward and outward over time. Anthers typically emerge from the upper floret of the pair first, while those from lower floret typically emerge later the same day or on following days. Spent anthers eventually drop from the tassel and are sometimes mistaken for the pollen when observed on the leaves or ground.


The yellow or white “dust-like” pollen that falls from a tassel represents millions of individual, nearly microscopic, spherical, yellowish- or whitish translucent pollen grains. Estimates of the total number of pollen grains produced per tassel range from 2 to 25 million. Each pollen grain contains the male genetic material necessary for fertilizing the ovary of one potential kernel.
The outer membrane of a pollen grain is very thin. Once dispersed into the atmosphere, pollen grains remain viable for only a few minutes before they desiccate. Yet, with only a 15 mph wind, pollen grains can travel as far as 1⁄2 mile within those couple of minutes.
Therein lies the concern of the potential for pollen “drift” from a transgenic corn field to an adjacent non-transgenic corn field and the risk of transgenic “contamination” of grain intended for non-transgenic markets. The good news is that recent research suggests that the overwhelming majority of a corn field’s pollen load is shed in the field itself.

All of the pollen from a single anther may be released in as little as three minutes. All the anthers on an individual tassel may take as long as seven days to finish shedding pollen, although the greatest volume of pollen is typically shed during the second and third day of anther emergence. Because of natural field variability in plant development, a whole field may take as long as 14 days to complete pollen shed.
Peak pollen shed usually occurs in mid-morning. Some research indicates that pollen shed decreases after temperatures surpass 86F. A second “flush” of pollen often occurs in late afternoon or evening as temperatures cool. Pollen shed may occur throughout most of the day under relatively cool, cloudy conditions.
Weather conditions influence pollen shed. If the anthers are wet, the pores will not open and pollen will not be released. Thus, on an average Indiana summer morning following a heavy evening dew, pollen shed will not begin until the dew dries and the anther pores open. Similarly, pollen is not shed during rainy conditions. Cool, humid temperatures delay pollen shed, while hot, dry conditions hasten pollen shed.
Extreme heat stress (100°F or greater) can kill corn pollen, but fortunately the plant avoids significant pollen loss by virtue of two developmental characteristics. First of all, corn pollen does not mature or shed all at once. Pollen maturity and shed occur over several days and up to two weeks. Therefore, a day or two of extreme heat usually does not affect the entire pollen supply. More importantly, the majority of daily pollen shed occurs in the morning hours when air temperature is much more moderate.
Related References
Kling, Jennifer G. and Gregory Edmeades. 1997. Morphology and growth of maize. IITA/CIMMYT Research Guide 9. Int’l Institute of Tropical Agriculture. [On-Line}. Available at <http://www.iita.org/cms/details/trn_mat/irg9/irg9.htm>(URL verified 7/2/07).
Nielsen, R.L. (Bob). 2007a. A Fast & Accurate Pregnancy Test for Corn. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timeless/EarShake.html>(URL verified 7/2/07).
Nielsen, R.L. (Bob). 2007b. Silk Emergence. Corny News Network, Purdue Univ. [On-Line]. Available at <http://www.kingcorn.org/news/timele
Ritchie, S.W., J.J. Hanway, and G.O. Benson. 1993. How a Corn Plant Develops. Iowa State Univ. Sp. Rpt. No. 48. [On-Line]. Available at <http://www.extension.iastate.edu/pages/hancock/agriculture/
corn/corn_develop/CornPlantStages.html>. (URL verified 7/2/07).
Russell, W.A. and A.R. Hallauer. 1980. Corn. (a chapter in) Hybridization of Crop Plants. American Soc. of Agronomy-Crop Science Soc. of America. Madison, WI.